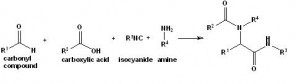
Topics containing organic chemistry and chemical reactions
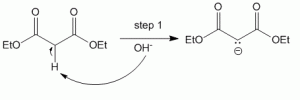
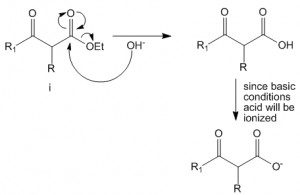
Abstraction of alpha-proton yields a carbanion which is stabilized by enol formation through resonance on either side of malonic ester
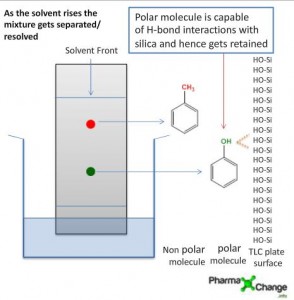
Malonic Ester Synthesis – Alkylation of Enolates
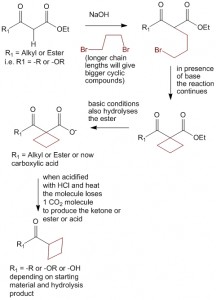
Using the non-selective nature of acetoacetic ester synthesis and malonic ester synthesis to produce cyclic compounds
Using the non-selective nature of acetoacetic ester synthesis and malonic ester synthesis to produce cyclic compounds
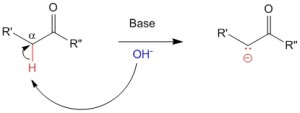
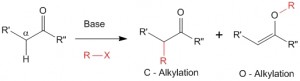
In the presence of a base alpha-hydrogens to carbonyl functional group can be abstracted very easily to yield a carbanion
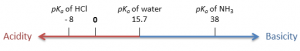
Chemistry of Enolates and Enols – Acidity of Alpha-Hydrogens
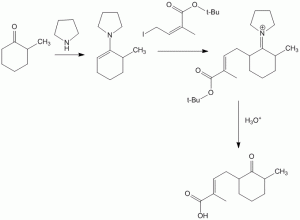
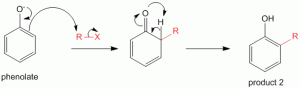
Reaction at the less reactive 6-position of 2-methyl cyclohexanone is forced due to formation of enamine
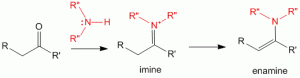
Producing and Using Imines and Enamines
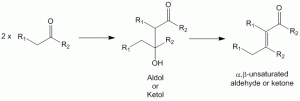
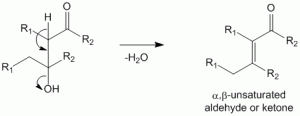
Aldol condensation - leading to aldol or ketol which can undergo dehydration to produce the alpha,beta-unsaturated aldehyde or ketone
Aldol Condensation – Acid Catalyzed
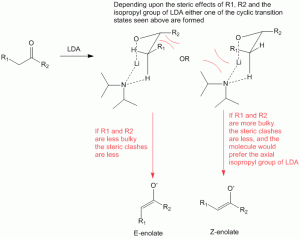
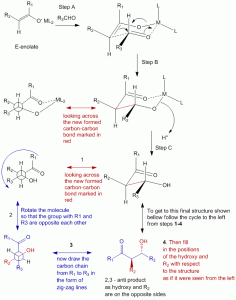
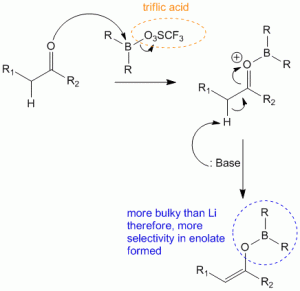
Formation of a cyclic transition state during enolate formation which controls formation of E-enol or Z-enol