Aldol synthesis gives rise to stereocenters in the molecule. Directed aldol synthesis is an approach in order to control the stereochemistry of the product being formed. Formation of the enolate is the first step in aldol synthesis, and hence stereocontrol must begin with the first step itself.
When a base reacts with a carbonyl compound, it can abstract the α-hydrogens to produce either a E-enolate or a Z-enolate. Assuming that the base is lithium diisopropyl amine (LDA), let us take a look at what happens to form the enolates for a generic carbonyl compound.
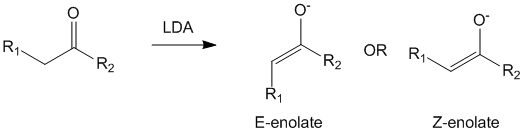
The control of whether this reaction produces the E-enolate or the Z-enolate mainly depends upon the bulkiness of the R1 and the R2 groups. This selectivity is imparted as per the Ireland model. The reason for this lies in the cyclic transition state that is formed in the production of these enols as can be seen below.
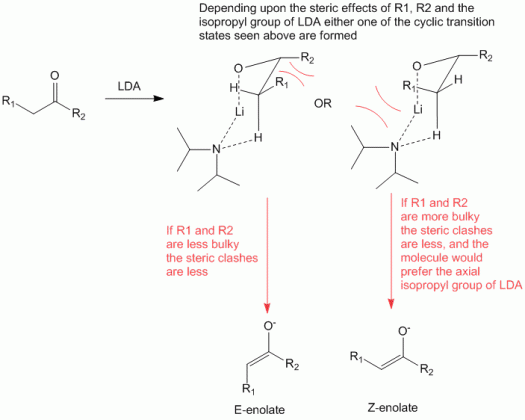
Thus as can be observed from the figure above which is a representative of the Ireland model:
- The LDA forms a cyclic transition state, such that the metal Lithium (Li) forms a coordinate bond with the oxygen, while the amine (diisopropyl amine) attempts to abstract the α-hydrogen of the carbonyl compound.
- If R1 and R2 are less bulky, the steric clashes between them are smaller, and hence they can be accommodated in the first transition state to produce the E-enolate.
- If R1 and R2 are more bulky, the steric clashes between them are very large, pushing the R1 group into the axial position (which is relatively disfavored due to 1,3-diaxial interactions). However, due to the bulkiness of the R1 and R2 groups, these axial interactions would be lower in comparison allowing this cyclic transition state to dominate to produce the Z-enolate.
How To Obtain Selectivity for E-Enolate
To obtain E-enolate for compounds having bulky and branched R1 and R2 groups the following steps can be done:
- Change the metal used in the base to one which would form lesser coordinate bonds. That way a cyclic transition state would be avoided.
- Change the solvent to a more polar solvent which would solvate the enolate formed, thereby preventing the formation of the cyclic transition state.
Books on Organic Chemistry
Check out some of the best textbooks for Organic Chemistry
References
- Advanced Organic Chemistry: Reactions and synthesis. By Francis A. Carey, Richard J. Sundberg
- R. E. Ireland, P. Wipf, J. D. Armstrong III; Stereochemical control in the ester enolate Claisen rearrangement. 1. Stereoselectivity in silyl ketene acetal formation. J. Org. Chem. 1991, 56, 650-657.

This article was very helpful to me. This site gives quality information. Would like to have more information.