Click Here To Get Multicomponent Reactions – Powerpoint Presentation
Abstract:
An ideal chemical reaction should be simple to perform, give maximum yields, utilize readily available starting materials, be a one-pot reaction and be environmentally friendly [1]. No reaction comes close to achieving this ideality; however, multicomponent reactions are best positioned to achieve this. By definition, multicomponent reactions are those reactions whereby more than two reactants combine in a sequential manner to give highly selective products that retain majority of the atoms of the starting material [2]. This makes multicomponent reactions (MCRs) highly convergent reactions, with high atom efficiency, good bond forming capabilities and higher yields in comparison to a similar multistep reaction.
Multicomponent reactions (or Multi-Component Reactions) have been known for over 150 years. The first documented multicomponent reaction was the Strecker synthesis of alpha-amino cyanides in 1850 [2] from which alpha-amino acids could be derived. A multitude of MCRs exist today, of which the isocyanide based MCRs are the most documented. Other MCRs include free-radical mediated MCRs, MCRs based on organoboron compounds and metal-catalyzed MCRs.
Isocyanide based MCRs are most frequently exploited because the isocyanide is an extraordinary functional group. It is believed to exhibit resonance between its tetravalent and divalent carbon forms [3]. This induces the isocyanide group to undergo both electrophilic and nucleophilic reactions at the CII atom, which then converts to the CIV form in a exothermic reaction. The occurrence of isocyanides in natural products has also made it a useful functional group. The two most important isocyanide-based multicomponent reactions are the Passerini 3-component reaction to produce alpha-acyloxy carboxamides and the Ugi 4-component reaction, which yields the alpha-acylamino carboxamides.
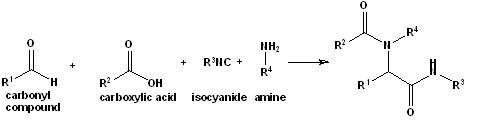 Figure 1. The Ugi 4-component reaction involving a carbonyl compound, a carboxylic acid, an isocyanide and an amine to yield ?-acylamino carboxamides.
Figure 1. The Ugi 4-component reaction involving a carbonyl compound, a carboxylic acid, an isocyanide and an amine to yield ?-acylamino carboxamides.
Multicomponent reactions are widely used in the synthesis of natural products to produce complex scaffolds. The earliest natural products synthesized were amino acids. However over time, with the development of new MCRs, more complex natural products could also be synthesized. For example, the Passerini reaction has found utility in the synthesis of azinomycin – a DNA binding and alkylating antibiotic [4], and eurystatin A- a natural prolyl peptidase inhibitor [5]. The Ugi reaction has found application in the synthesis of antibiotic bicyclomycin [6] and the synthesis of the toxin dysidenin [7]. The Ugi reaction has also been recently exploited to synthesize 2-oxazolines, which are known to be important pharmacophores in bioactive molecules [8]. A two step synthesis towards substituted benzoxazinones has also been reported by performing the Ugi reaction followed by an intramolecular Mitsunobu substitution [9].
Apart from changing substituents on the reacting components, modifications of the individual component functional groups of an MCR allows the generation of more scaffolds using almost similar reaction conditions. An example of such a modification is the replacement of the amine in the Ugi reaction with an O-benzyl hydroxamine which following the Ugi reaction can be easily deprotected to the hydroxamic acids [10]. These hydroxamic acids can be useful to develop enzyme inhibitors. Cyclic variations of the MCRs are also possible in which two or more components could be connected by a chain to produce heterocycles. For example, the Ugi reaction has been used to synthesize ?-lactam rings [11].
Multicomponent reactions are not just an integral part of synthesizing natural products, but due to their ability to produce a variety of scaffolds by simple modifications of substituents, they are being exploited for the discovery of new drugs as well. Novel inhibitors of enzymes such as serine proteases (Factor Xa) have been synthesized using MCRs [12]. Tubulin inhibitors, which could potentially be used as anticancer agents, have also been synthesized using a van Leussen 3 component reaction [13]. Development of new antimalarial drugs based on the chloroquine scaffold has also been researched [14]. Additional uses of MCR in drug discovery have been reviewed [15].
A new concept to increase the potential variety in MCRs is the union of MCRs. In this combination of two or more known MCRs is done such that the end product of one MCR acts as the starting product of the other MCR. A 7-component MCR has been developed which combines the Asinger 4-component reaction and the Ugi 4-component reaction [16]. Clearly the utility of MCRs is gaining importance and wider acceptance from drug development to synthesis of natural products. The future requires increased utilization of the already present MCRs as well as development of novel MCRs and the union of MCRs to achieve more efficient synthesis.
Books on Multi-Component Reactions
Check out some really good books on Multi-component Reactions
References
- Wender, P. A.; Handy, S.; Wright, D. L. Towards the ideal synthesis. Chem. Ind. 1997, 765-774.
- Domling, A; Ugi, I. Multicomponent Reactions with Isocyanides. Angew. Chem. Int. Ed. 2000, 39, 3168-3210.
- Ugi, I.; Fetzer, U.; Eholzer, U.; Knupfer, U.; Offermann, K. Isonitrile Syntheses. Angew. Chem. Int. Ed. 1965, 4, 474-484.
- Armstrong, R. W.; Moran, E. J. Highly Convergent Approach to the Synthesis of the Epoxy-Amide Fragment of the Azinomycins. Tetrahedron Lett. 1991, 32, 3807-3810.
- Schmidt, U.; Weinbrenner, S. The Synthesis of Eurystatin A. J. Chem. Soc., Chem. Commun. 1994, 1003.
- Fukuyama, T.; Robins, B. D.; Sachleban, R. A. Synthetic Approach to Bicyclomycin: Synthesis of the Bicyclic System of Biclomycin. Tetrahedron Lett. 1981, 27, 4155-4158
- De Laszlo, S. E.; Williard, P. G. Total synthesis of (+)-demethyldysidenin and (-)-demethylisodysidenin hexachlorinated amino acids from the marine sponge Dysidea herbacia. Assignment of absolute stereochemistry. J. Am. Chem. Soc. 1985, 107, 199-203.
- Fan, L.; Lobkovsky, E.; Ganem, B. Bioactive 2-oxazolines: A New Approach via One-Pot, Four Component Reaction. Org. Lett. 2007, 9, 2015-2017.
- Banfi, L.; Basso, A.; Guanti, G.; Lecinska, P.; Riva, R. Multicomponent synthesis of benzoxazinones via tandem Ugi/Mitsunobu reactions: an unexpected cine-substitution. Mol. Divers. 2008, 12, 187-190.
- Basso, A.; Banfi, L.; Guanti, G.; Riva, R.; Riu, A. Ugi multicomponent reaction with hydroxylamines: an efficient route to hydroxamic acid derivatives. Tetrahedron Lett. 2004, 45, 6109-6111.
- Ugi, I. From Isocyanides via Four-Component Condensations to Antibiotic Syntheses. Angew. Chem. Int. Ed. Engl. 1982, 21, 810-819.
- Nerdinger, S.; Fuchs, T.; Illgen, K.; Eckl, R. Aryl amides that inhibit factor Xa activity. WO 2002068390, September 6, 2002.
- Wang, L.; Woods, K. W.; Li, Q.; Barr, K. J.; McCroskey, R. W.; Hannick, S. M.; Gherke, L.; Credo, R. B.; Hui Yu-Hua; Marsh, K.; Warner, R.; Lee, J. Y.; Zielinski-Mozng, N.; Frost, D.; Rosenberg, S. H.; Sham, H.L. Potent, Orally Active Heterocycle-Based Combretastatin A-4 Analogues: Synthesis, Structure-Activity Relationship, Pharmacokinetics, and In Vivo Antitumor Activity Evaluation. J. Med. Chem. 2002, 45, 1697-1711.
- Musonda, C. C.; Taylor, D.; Lehman, J.; Gut, J.; Rosenthal, P. J.; Chibale, K. Application of multi-component reactions to antimalarial drug discovery. Part1: Parallel synthesis and antiplasmodial activity of new 4-aminoquinoline Ugi adducts. Bioorg. Med. Chem. Lett. 2004, 14, 3901-3905.
- Domling, A. Recent Developments in Isocyanide Based Multicomponent Reactions in Applied Chemistry.Chem. Rev. 2006, 106, 17-89.
- Domling, A.; Ugi, I. The Seven-Component Reaction. Angew. Chem. Int. Ed. Engl. 1993, 32, 563-564


The site is worth to read. The uniqueness and balance that reflects from this article. Now-a-days blogs are used in each and every field. The knowledge that we recieve from them has no words to describe. The attribute required is the power of creativity within yourself via learning, thinking, creating and rigorous study. Therefore the article is truely fruitful for the readers. Thank you a lot for providing such an awesome article. I await your future article with great egarness.