When a carbonyl containing compound (such as aldehydes and ketones) reacts with an amine (primary, secondary or ammonia), it yields the formation of imines which in turn get converted to enamines.
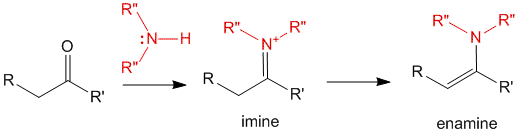
The enamine form of the product is analogues to the enolate form of the carbonyl compound. The advantages of this enamine will be highlighted down below. But first let us take a look at the mechanism of formation of these imines and enamines.
NOTE- imines and enamines are tautomers just like carbonyl compounds and enolates.
Mechanism
Step 1
In step 1, the amine with its lone pair of electrons attacks the carbonyl carbon directly to yield a zwitterionic intermediate which then undergoes a tautomerism to yield the alcohol amine.
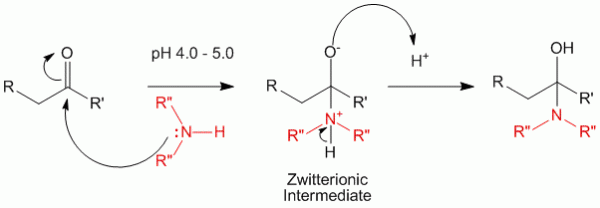
It is important to note that this reaction happens in slightly acidic conditions an therefore a pH of 4.0 to 5.0 is preferred.
Step 2
The alcohol of the alcohol amine then attacks the proton in the acidic conditions to release the water molecule yielding the formation of the imine.
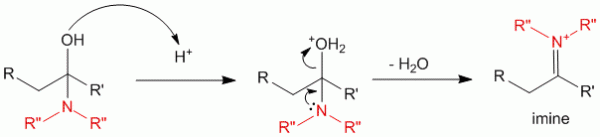
Step 3
The imine being a charged species would quickly undergo a resonance transformation to form the enamine.
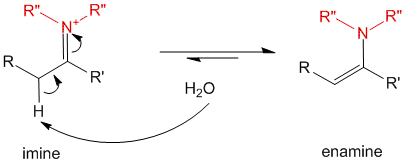
Under some circumstances, the imine may be stable enough and hence an equilibrium may exist between the two forms depending on the substituents on the molecule.
Activation of α-Carbon of Carbonyl Compounds
The enamine is just like an enol. The nitrogen of the enamine can donate its lone pair into the chain to produce the carbanion at the α-carbon. The formation of the carbanion in case of enamines is easier when compared to enols, because the nitrogen which donates the lone pair is less electronegative as compared to the oxygen in enols. Thus formation of enamine can help in activating carbonyl compounds to reactions at the α-carbons.
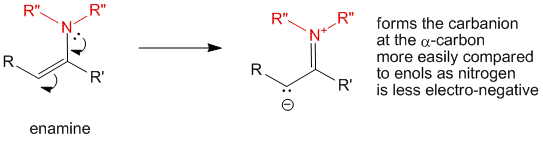
Formation of enamine is a useful method of activation of less or non-reactive ketones. It is also useful in performing reactions of enols under milder conditions.
Books on Organic Chemistry
Check out some of the best textbooks for Organic Chemistry
References
- Advanced Organic Chemistry: Reactions and synthesis. By Francis A. Carey, Richard J. Sundberg
- Organic Chemistry, 6th Edition. By Robert T. Morrison and Robert N. Boyd