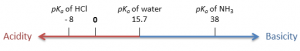
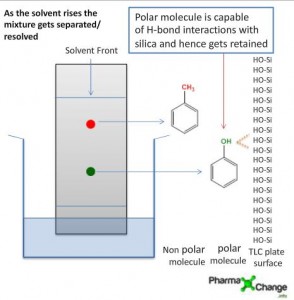
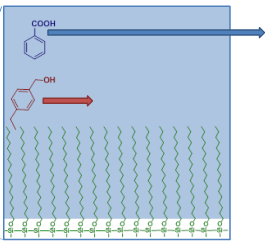
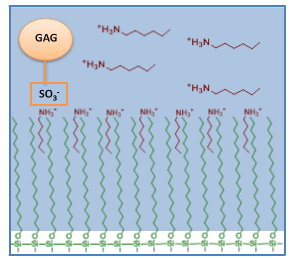
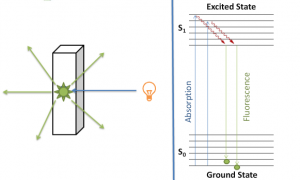
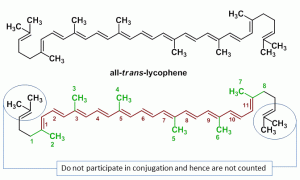
Notes to understand topics in analytical chemistry.
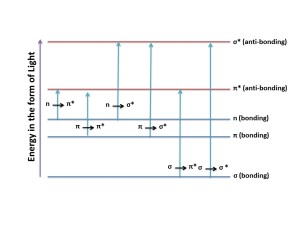
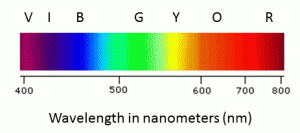
Different transitions between the bonding and anti-bonding electronic states when light energy is absorbed in UV-Visible Spectroscopy.
Ultraviolet-Visible (UV-Vis) Spectroscopy – Principle
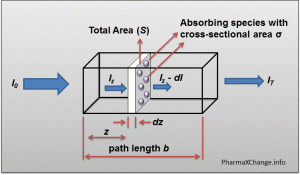
A diagrammatic representation of the hypothetical situation to derive the Beer-Lambert law containing a sample of path length b through which monochromatic light is passed.
Ultraviolet-Visible (UV-Vis) Spectroscopy – Derivation of Beer-Lambert Law
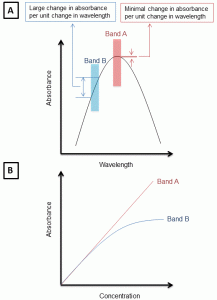
Figure A: Shows the difference in deviations in absorbance when values are obtained at maximum wavelength of absorbance (band A) vs other wavelengths of absorbance (band B). Figure B: shows the deviations in Beer-Lambert law due to observations made at wavelengths other than lambda max.