Introduction
While the ill-effects of cholesterol are widely known, most people are unaware that cholesterol plays multiple important roles in the human body such as being a precursor for formation of steroid hormones, bile acids, lipoproteins (the form in which lipids are transported in the body) and vitamin D. Excessive cholesterol is associated with several cardiovascular diseases and such levels are easily attained due to unhealthy diet. In fact, it is not essential for cholesterol to be obtained from the diet as it is easily synthesized in the body. Our body produces almost 1g of cholesterol per day. Let us therefore take a closer look into this biological pathway.
Biosynthesis of Cholesterol
Cholesterol is biosynthesized in various tissues of the human body. Cytosome and mitochondria of the cells of these tissues contain the enzymes required to carry out the synthesis of cholesterol. The intestine and liver synthesize the most cholesterol out of all the organs. Cholesterol synthesis was studied by feeding animals with acetate labeled at either the methyl or the carboxyl carbon with radiolabelled 14C. The data obtained from this helped to make a guideline for cholesterol synthesis and concluded that all the carbon atoms in cholesterol are derived from acetate.
It is estimated that synthesis takes place in four stages as described in the animation below.
Press the play button to move ahead in the animation in a step-wise manner. The description of the entire process is given in detail following the animation.
[swfobject]2824[/swfobject]
Here is a detailed description of the four stages:
-
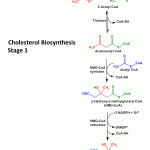
Cholesterol Biosynthesis – Stage 1 Formation of mevalonate (6C) by the condesation of three acetate (2C) molecules –
The chain of reaction starts with the condensation of 2 acetate molecules to form acetoacetyl-CoA in the presence of the enzyme thiolase.
This acetoacetyl-CoA then reacts with one more molecule of acetate catalysed by HMG-CoA synthase to give the six-carbon compound β-hydroxy-β-methylglutaryl CoA (HMG-CoA).
Now this six carbon compound undergoes reduction by HMG-CoA reductase to form mevalonate. This is a rate limiting step and the electrons are donated by 2 molecules of NADPH. The cholesterol lowering drugs called “statins” such as Atorvastatin inhibit HMG-CoA reductase in order to lower the body’s production of cholesterol.
-
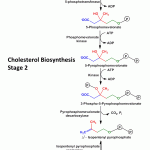
Cholesterol Biosynthesis Stage 2 Formation of activated isoprene (5C) units ( Δ3-Isopentenyl pyrophosphate and dimethylallyl pyrophosphate) from mevalonate –
Kinases catalyse the addition of phospahte groups to mevalonate to form 3-phospho-5-pyrophosphomevalonate.
3-phospho-5-pyrophosphomevalonate by releasing carbon dioxide forms the first activated isoprene unit Δ3– isopentenyl pyrophosphate. The phosphate groups are donated by 3 molecules of ATP.
The second activated isoprene unit dimethylallyl pyrophosphate is obtained by isomerisation of Δ3-isopentenyl pyrophosphate.
-
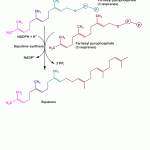
Cholesterol Bosynthesis Stage 3… contd. 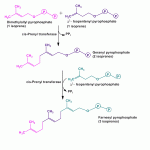
Cholesterol Biosynthesis Stage 3 Formation of squalene (30C) (6-isoprenes) by polymerisation of activated isoprene units–
The previously formed isoprene units undergo a head-to-tail condensation to form geranyl pyrophosphate by displacing one pyrophosphate group.
This 10 carbon compound then reacts with one more molecule of isopentenyl pyrophosphate to yield a 15 carbon compound called as farnesyl phosphate.
Finally squalene is formed with the condensation of 2 farnesyl molecules in a head-to-head manner by the displacement of both the pyrophosphate groups.
-
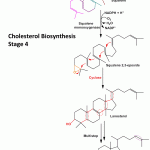
Cholesterol Biosynthesis Stage 4 Formation of cholesterol by cyclization of squalene (to form the sterol nucleus) and a number of other reactions-
An oxygen atom is added to the end of the squalene chain by squalene monooxygenase to form squalene 2,3 epoxide. The other oxygen atom is reduced by NADPH to water.The oxygen atom attached forms a epoxide ring, which along with the double bonds in the structure of squalene 2,3 epoxide is unique to carry out cyclization of the linear structure of squalene. Cyclization by cyclase to form lanosterol.
This lanosterol on undergoing further a series of reactions (~ 20) involving removal and addition of certain methyl groups forms cholesterol as highlighted in the figure. 7-dehydrocholesterol, a precursor to vitamin D3, is the penultimate product formed before the formation of cholesterol.
The cholesterol formed now has many fates. A minor portion of the cholesterol formed in the liver is incorporated into the liver cell membranes while the rest is transported in the form of bile acids (fecal excretion) or cholesteryl esters.
Regulation of Cholesterol Biosynthesis
Under certain conditions the cholesterol obtained from the diet as well as the cholesterol synthesized in the body in combination exceed the amount required by the body for production of steroids, cellular membranes or biles. This leads to accumulation of the excess cholesterol in the blood vessels. This accumulated cholesterol causes plaques which can result in various cardiovascular diseases. As excessive cholesterol can be injurious to health, regulation of synthesis of cholesterol in the body has to be maintained. Moreover cholesterol synthesis is an energy consuming and a complex process thus the process should be well controlled and monitored by the body naturally.
- Cholesterol synthesis is controlled by certain hormones like glucagon and insulin but the main step that regulates cholesterol synthesis is the conversion of HMG-CoA to mevalonate in presence of HMG-CoA reductase. This enzyme HMG-CoA reductase is thus the rate limiting enzyme and controls excessive cholesterol formation by feedback mechanism.
When the concentration of intracellular cholesterol increases, levels of HMG-CoA reductase are reduced by decreasing the gene transcription of this enzyme. This mechanism was concluded when studies were done to investigate LDL (low density lipoprotein) level regulation. - As mentioned above the enzyme HMG-CoA reductase is also regulated by hormones to regulate cholesterol synthesis. Covalent modification of the enzyme itself causes activation/deactivation of the enzyme and increase/decrease in levels of hormonal release. The phosphorylated form of HMG-CoA reductase is the inactivated state, whereas the the dephosphorylated form is in the active state. Dephosphorylation is promoted by insulin thereby activating the enzyme and in turn increasing cholesterol synthesis whereas phosphorylation is triggered by glucagon thereby deactivating the enzyme and in turn reducing cholesterol synthesis. These hormones exert their effects through a series of reactions via cAMP.
Also, when concentrations of cholesterol is high in the cells, an enzyme called acyl CoA-cholesterol acyl transferase is released which elevates esterification of cholesterol for storage. Moreover high cholesterol level sends a signal to the gene encoding LDL receptors to decreases transcription and in turn reduce production of receptors. These causes a fall in level in uptake of cholesterol from the blood. - When cholesterol in the body, due to genetic disorder, continues to be produced despite the excess cholesterol in the blood (lack of uptake by LDL receptor) it causes hypercholesterolemia. In such conditions, drugs like atorvastatin, compactin and lovastatin are used to regulate cholesterol levels.They act by inhibiting the enzyme HMG-CoA reductase and in turn reduced production of cholesterol.
Recommended Texts
- David L. Nelson and Michael M. Cox, Lehninger Principles of Biochemistry 6th Edition
- Jeremy M. Berg, John L. Tymockzo and Luber Stryer, Biochemistry 7th Edition
- Reginald H. Garrett, Charles M. Grisham, Biochemistry by Reginald H Garrett 5th Edition
.
- U. Satyanarayana, U. Chakrapani, Biochemistry by U. Satyanarayana. 3rd Edition.