As we have seen in Pharmacokinetics Basics-Absorption, Distribution, Metabolism and Excretion that each of these process have a significant role to play in our body. Metabolism plays a vital protective function within the body by converting pharmaceutical drugs and xenobiotics into easily excretable hydrophilic compounds. In this article we discuss the significance of metabolism and a general overview of the mechanisms by which our body performs metabolism.
Significance of Metabolism
- Most of the drugs are lipophilic in nature, thus easily absorbed through the gastrointestinal (GI) lipoproteins membranes. Once absorbed in the blood, these drugs reach their target tissues to explicit their pharmacological activities. Once they have shown their action, these drugs have to be excreted out of the body. Lipophilic content cannot be excreted via urine. Thus, via metabolism, these drugs by action of specific enzymes are converted to their hydrophilic forms which can then be easily excreted in the urine. If the drugs are not metabolized to their water soluble excretable form, they would remain in the body for an uncertain amount of time explicating their biological effects.
- Metabolism plays an important role not only in excretion of drugs but also foreign compounds/xenobiotics such as when natural toxins/pesticides are ingested from fruits and vegetables, carcinogenic products are formed during charbroiling of meat, ingestion of drinks or smoking, inhalation of volatile chemicals etc. Metabolism protects the body from harmful xenobiotics rendering them relatively inactive and non toxic. Thus metabolism is considered as a detoxification process (as for most of the cases).
- However, at certain times, the procarcinogens (eg. benzo [α]pyrene) via metabolism can get converted to their active metabolite in the body, thus rendering harmful effects. These active metabolites may covalently bind with proteins or DNA leading to carcinogenicity or mutagenicity. In such cases metabolism does not play a detoxifying role, but instead results in the formation of toxic compounds.
- Inactive drugs which get converted to their active metabolites in the body are known as prodrugs. Prodrugs can be useful in creating more bioavailable drugs which can be readily absorbed by the body and show their action after metabolism thereby prolonging the duration of action. An example of a prodrug is Dabigatran- a thrombin inhibitor.
- Also drugs are developed with the concept that ‘one dose for all’ not considering the fact that all human beings are metabolically and genetically different, thereby giving variable responses. Accounting for the metabolism mechanisms and the condition of the patient’s metabolism can be useful in making informed decisions about dosing in therapy.
- Moreover for people taking long term therapy such as in cases of cardiovascular diseases or cancer etc problems of drug toxicity may occur. Drug accumulation may occur due to faulty metabolism or excretion in a patient and therefore knowing the metabolic pathways could possibly prevent toxicity in therapy.
- When several drugs are prescribed for a single individual, knowledge of the metabolic effects of the drugs can prevent drug-drug interactions or even drug-food interactions can prevent adverse effects.
- As a drug is taken orally, it is absorbed from the GI tract and it passes through the liver before it is distributed to all the tissues in the body. Certain drugs undergo metabolism either by the liver cells or extra hepatic tissues before they reach the systemic circulation forming either inactive or active (eg. prodrugs). This is known as first pass metabolism or presystemic first pass effect. eg. Lidocaine when given orally, gets ineffective because of the first pass metabolism. Nitroglycerin is administered buccaly to avoid first pass metabolism. Understanding mechanism of metabolism is important to therefore determine the ideal route of administration.
Detoxification or the toxic effects (eg. procarcinogens) of metabolism vary in human beings due to a number of factors such as genetic difference between different CYP-450 enzyme isoforms, hormones, gender, age, disease, and environmental factors. Hence a good understanding of metabolism is very important from a pharmacists point of view.
How our Body Carries Out Metabolism of Drugs and Xenobiotics
The main site for metabolism in the body is the liver, but intestinal mucosa also carries out metabolism for some orally administered drugs. Cytochrome P-450 enzymes (and its isoforms) are generally involved to carry out metabolism and are present in the endoplasmic reticulum of the cells. There are other sites where metabolizing enzymes are also found such as kidney, lung, nervous tissue etc. (Eg. of drugs getting metabolised by the intestinal mucosa are isoproterenol, levodopa, chlorpromazine etc.)
Our body carries out metabolism in two phases (as described earlier in Pharmacokinetics basics) generally sequentially:
Phase I (functionalization or biotransformations)- involves the addition of a new functional group in the substrate molecule, or the existing functional group is modified or unmasked or either the functional group or the phase II reactions acceptor site is exposed.
Polar groups such as OH, COOH, SH, NH2 are added. Reactions included in this phase are oxidation, hydroxylation, reduction and hydrolysis. This is done to make the compound more water soluble.
Although addition of these functional groups does not make the compound sufficiently hydrophilic in this phase for excretion. The polar groups added in phase I act as handles for phase II of the metabolism and hence the compound is prepared/made ready in this phase for it to subsequently undergo phase II conjugation reactions to enhance its hydrophilicity.
Phase II (conjugation)- involves the masking of the existing functional group with addition of a new group in the presence of enzymes to make the drug/metabolite more hydrophilic so that it can be easily excreted.
Here a small polar group is added to the metabolites of phase I reactions or to the parent compounds which already have suitable existing functional groups to make them more water soluble. Groups such as glucuronic acid, glycine, sulfate or amino acids are added. Other phase II reactions such as methylation and acetylation make the molecule inactive, whereas glutathione conjugates protects the body from the active/chemically reactive metabolites.
Certain drugs/xenobiotics which are already hydrophilic enough or are insusceptible to the metabolizing enzymes get excreted unchanged.
The following metabolic reactions are discussed further in detail:
Phase I
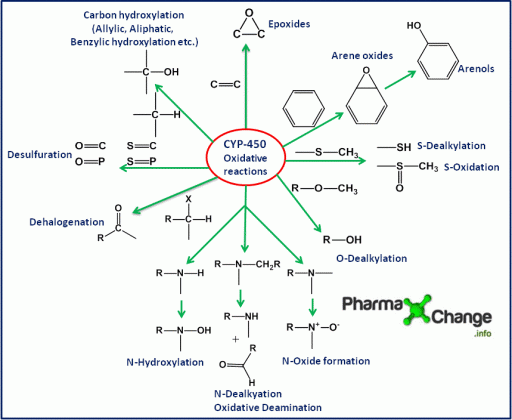
::Oxidative reactions::
- Oxidation of aromatic compounds
- Oxidation of aliphatic and alicyclic compounds
- Oxidation of alkenes and alkynes
- Oxidation of benzylic, allylic carbon atoms and carbon atoms α to carbonyl and imines
- Oxidation of alcohols and aldehydes
- Oxidation involving carbon-heteroatom systems:
- Carbon-nitrogen systems including aliphatic and aromatic amines (N-dealkylation, oxidative deamination, N-oxidation, N-hydroxylation)
- Carbon-oxygen systems (O-dealkylation)
- Carbon-sulphur systems (S-dealkylation, S-oxidation, Desulfuration)
- Carbon-halogen systems (Dehalogenation)
::Reductive reactions::
- Reduction of aldehydes and ketones
- Reduction of nitro and azo compounds
::Hydrolytic reactions::
- Hydrolysis of esters and amides
- Hydrolysis of epoxides and arene oxides
Phase II
- Glucuronic acid conjugation
- Sulfate conjugation
- Conjugation with amino acids
- Glutathione or Mercapturic acid conjugation
- Acetylation
- Methylation
References:
1. Foye’s Principles of Medicinal Chemistry (Buy at Amazon.com)
2. Wilson & Grisevold’s Textbook of Organic Medicinal Chemistry. (Buy at Amazon.com)