Click Here To Get Presentation
Abstract
Isothermal titration calorimetry (ITC) is a technique used to measure the heat exchange associated with molecular interactions at constant temperature, most often as a direct method for finding thermodynamic parameters associated with complex formation.1,2 It provides a complete thermodynamic profile in a single experiment, including enthalpy changes (ΔH), binding affinity/association constant (Ka) and stoichiometry (n).3,4 From these initial measurements, changes in entropy (ΔS) and Gibbs free energy (ΔG) can be determined using the equation: ΔG = -RTlnKa = ΔH – TΔS. 5,6 These parameters are related to the mechanism of binding to a biomolecule. Therefore, it is the most preferred method for measuring the thermodynamics of ligand binding.5
In 1965, Christensen et al first described titration calorimetry as a method for simultaneous determination of equilibrium constant and enthalpy.7 This technique was later used for binding studies in biological systems.2 In 1989, one of the first commercial titration calorimeters was made available. Since then ITC is routinely used to directly characterize the thermodynamics of biopolymer binding interactions. In 1993, this technique was used to characterize kinetic parameters for enzymes.2,8 Currently there are four ITC instruments available on the market: VP–ITC, iTC200, Auto–iTC200 and NANO ITC. Of all these Auto–iTC200 is the most widely used.6
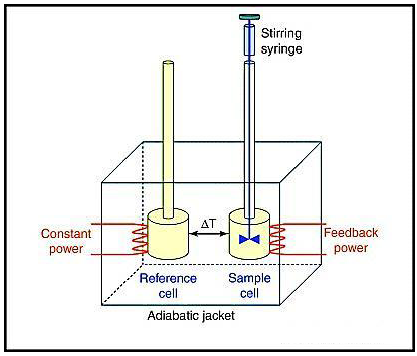
An isothermal titration calorimeter consists of two cells, a sample cell and a reference cell, made up of efficient thermal conducting material, surrounded by an adiabatic jacket. A syringe is used to titrate the ligand into the sample cell containing the biomolecule. Sensitive thermopile or thermocouple circuits are used to detect temperature differences between the two cells, while maintaining isothermal conditions between them (ΔT=0) (Figure 1).9
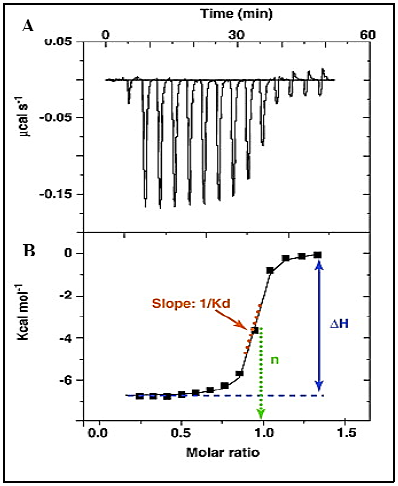
During the titration process, heat change in the sample cell is observed. The power required to maintain isothermal conditions between the two cells, is measured by a calorimeter.9 A plot of power (μcals-1) against time (min) shows a series of peaks of heat flow (power) (Figure 2A). The area under the peaks gives the enthalpy (ΔH) of binding.1,5 A binding isotherm obtained from the plot of heat of formation against the molar ratio of ligand to biomolecule (Figure 2B) gives information about the enthalpy (ΔH), the association constant (Ka) and the stoichiometry (n) of binding, which can be used to calculate changes in entropy (ΔS) and Gibbs Free Energy (ΔG).5,9
Isothermal Titration Calorimetry can measure Ka values between molecules ranging from millimolar to nanomolar range.10 Displacement or competitive binding methods need to be performed for ligands having binding affinities in nanomolar to picomolar range. In this method, a strong ligand is titrated with a mixture of biomolecule and weak ligand. The binding affinity of the strong ligand is found to decrease on competing with the weak ligand for the binding site.11,12
Isothermal Titration Calorimetry is fast becoming a major tool in drug discovery studies. It is typically used as a secondary screening technique, to eliminate false positive hits after primary screening. Moreover, it is utilized for hit selection, lead optimization and validation of computational estimates of binding energetics.3,13 It is also used in binding studies (drug–macromolecule, drug–DNA/RNA, protein–protein interactions), where the binding mechanism, forces involved in binding and strength of the interaction can be analyzed.11,14,15 In conjunction with chromatography, ITC can be used to identify and isolate unknown target proteins including receptors and cell/tissue lysates.16,17 ITC provides insights into the SAR for ligand interaction with the target.18,19 This information is imperative in drug design and lead optimization. Using structural and thermodynamic information of binding, lead compounds can be further optimized.4,20,21 ITC is also gaining popularity as one of the methods for studying enzyme kinetics. It provides invaluable data on the kinetics and thermodynamics of enzyme catalysis, the occurrence of inhibition and its corresponding mechanism.3,22,23
Isothermal Titration Calorimetry has advantages over other techniques such as fluorescence assays, NMR and SPR for studying complex formation in terms of ease of use and cost. It does not require any fluorescent probes or radioactive tags for data analysis. Immobilization and chemical modification of protein is not required.10,21 Also, it does not have limitations associated with clarity of the solution, molecular weight, temperature or pH. Nonetheless, low sensitivity, low throughput and large sample requirement are major concerns of this technique.11,16
Although the technique is not very sensitive, it is one of the best methods for determining the thermodynamic parameters of ligand binding. Improvements in the sensitivity of the instrument, decrease in the sample quantity required for titration and increases in the throughput capacity will make ITC a valuable technique in drug discovery.14
Books on Isothermal Titration Calorimetry
Check out some of the best books to find information on Isothermal Titration Calorimetry:
References
- Perozzo, R.; Folkers, G.; Scapozza, L. Thermodynamics of protein–ligand interactions: History, presence, and future aspects. J. Recept. Sig. Transd. 2004, 24, 1–52.
- Freyer, M. W.; Lewis, E. A. Isothermal titration calorimetry: Experimental design, data analysis, and probing macromolecule/ligand binding and kinetic interactions. Methods Cell Biol. 2008, 84, 79–113.
- Peters, W. B.; Frasca, V.; Brown, R.K. Recent developments in isothermal titration calorimetry label free screening. Comb. Chem. High T. Scr. 2009, 12, 772–790.
- Chaires, J. B. Calorimetry and thermodynamics in drug design. Annu. Rev. Biophys. 2008, 37, 135–151.
- Holdgate, G. A.; Ward, W. H. J. Measurement of binding thermodynamics in drug discovery. Drug Discov. Today 2005, 22, 1543–1550.
- Torres, F. E.; Recht, M. I.; Coyle, J. E. Higher throughput calorimetry: Opportunities, approaches and challenges. Curr. Opin. Cell Biol. 2010, 20, 598–605.
- Christensen, J. J.; Izatt, R. M.; Eatough, D. Thermodynamics of metal cyanide coordination. V. Log K, ΔHo, and ΔS values for the Hg2 + -cn-system. Inorg. Chem. 1965, 4, 1278–1280.
- Williams, B. A.; Toone, E. J. Calorimetric evaluation enzyme kinetics parameters. J. Org. Chem. 1993, 58, 3507–3510.
- Nunez, S.; Venhorst, J.; Kruse, C. G. Target–drug interactions: First principles and their application to drug discovery. Drug Discov. Today 2012, 17, 10–22.
- Edink, E.; Jansen, C.; Leurs, R. The heat is on: Thermodynamic analysis in fragment-based drug discovery. Drug Discov. Today: Technologies 2010, 7, 189–201.
- Liang, Y. Application of isothermal titration calorimetry in protein science. Acta Biochem. Biophys. Sin. 2008, 40, 565–576.
- Velazquez–Campoy, A.; Freire, E. Isothermal titration chromatography to determine association constants for high–affinity ligands. Nat. Protoc. 2006, 1, 186–191.
- Leavitt, S.; Freire, E. Direct measurement of protein binding energetics by isothermal titration calorimetry. Curr. Opin. Struc. Biol. 2001, 11, 560–566.
- Ghai, R.; Falconer, R.J.; Collins, B. M. Applications of isothermal titration calorimetry in pure and applied research–Survey of the literature from 2010. J. Mol. Recognit. 2012, 25, 32–52.
- De Azevedo, W. F.; Dias, R. Experimental approaches to evaluate the thermodynamics of protein–drug interactions. Curr. Drug Targets 2008, 9, 1071–1076.
- Zhou, X.; Sun, Q.; Kini, R. M.; Sivaraman, J. A universal method for fishing target proteins from mixtures of biomolecules using isothermal titration calorimetry. Protein Sci. 2008, 17, 1798–1804.
- Zhou, X.; Kini, R. M.; Sivaraman, J. Application of isothermal titration calorimetry and column chromatography for identification of biomolecular targets. Nat. Protoc. 2011, 6, 158–165.
- Sarver, R. W.; Peevers, J.; Cody, W. L. Binding thermodynamics of substituted diaminopyrimidine renin inhibitors. Anal. Biochem. 2007, 360, 30–40.
- Ferenczy, G. G.; Keseru, G. M. Thermodynamics guided lead discovery and optimization. Drug Discov. Today 2010, 15, 919–932.
- Velazquez-Campoy, A.; Luque, I.; Freire, E. The application of thermodynamic methods in drug design. Thermochim. Acta 2001, 390, 217–227.
- Ladbury, J. E.; Klebe, G.; Freire, E. Adding calorimetric data to decision making in lead discovery: A hot tip. Nat. Rev. Drug Discov. 2010, 9, 23–27.
- Noske, R.; Cornelius, F.; Clarke, R.J. Investigation of the enzymatic activity of the Na+, K+-ATPase via isothermal titration microcalorimetry. Biochim. Biophys. Acta 2010, 1797, 1540–1545.
- Todd, M. J.; Gomez, J. Enzyme kinetics determined using calorimetry: A general assay for enzyme activity?. Anal. Biochem. 2001, 296, 179–187.