Introduction:
As we discussed under metabolism of amino acids, ammonia produced is toxic to the body. Hence, if it is not reused to synthesize new amino acids or other nitrogen containing compounds, it is excreted out of the body as urea. This process of formation of urea occurs via the urea cycle for most terrestrial animals, also known as ureotelic species. There are exceptions to this, like aquatic species which excrete ammonia directly into the surroundings which gets diluted with the water and are called ammonotelic species. Also certain terrestrial species like birds and reptiles excrete ammonia as uric acid and hence are known as uricotelic species. The urea once produced, is excreted out by the kidneys in the urine. Urea is also called carbamide and when dissolved in water has a neutral pH.
Urea cycle
Ammonia is converted to urea in the hepatocytes of the liver in five steps via urea cycle- in the mitochondria (first 2 steps) and cytosol (last 3 steps). The urea then travels through the blood stream to the kidney and is excreted in the urine. The urea cycle was discovered by Hans Krebs (who also discovered Citric acid or Krebs cycle) and his student associate Kurt Henseleit in 1932. Urea cycle is discussed in detail as follows:
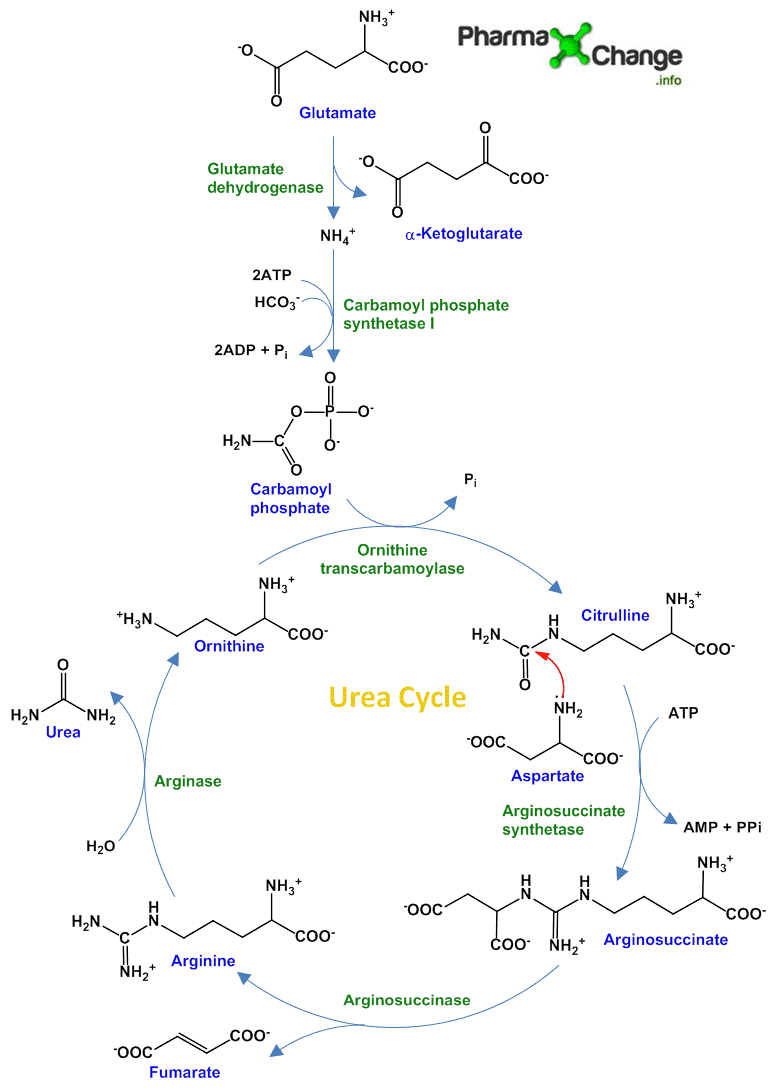
- Synthesis of carbamoyl phosphate– This step takes place in the mitochondria of the liver cells. Here the ammonium ions react with carbon dioxide (product of mitochondrial respiration) to form carbamoyl phosphate catalyzed by the enzyme carbamoyl phosphate synthetase I. This is an irreversible, rate-limiting, ATP-dependent reaction and consumes 2 ATP. Carbamoyl phosphate synthetase I (in mitochondria) is different from carbamoyl phosphate synthetase II (in cytosol) as the latter one has a different role to play and is involved in pyrimidine synthesis.
- Synthesis of citrulline– Carbamoyl phosphate produced in the first step reacts with ornithine in the presence of ornithine transcarbamoylase to synthesize citrulline. Just like oxaloacetate in Kreb’s cycle, ornithine plays a similar role acting as accepting substrate at each turn of the cycle. Via a transporter system this citrulline is now transferred to the cytosol of the liver cells.
- Formation of arginosuccinate– In this ATP dependent step, the carbonyl carbon of citrulline is attacked by the lone pair of the amine in aspartate to produce arginosuccinate in presence of arginiosuccinate synthetase. In this step, the second nitrogen of urea is incorporated by condensation. ATP is broken down into AMP and pyrophosphate.
- Breakdown of arginosuccinate– Arginosuccinase promotes the cleavage of arginosuccinate to give arginine and fumarate in a reversible manner. Fumarate formed here joins the citric acid cycle forming a link between urea and citric acid cycle.
- Formation of urea– Arginine produced in the earlier step is broken down by arginase to give urea and ornithine. Ornithine is recycled back to the mitochondria for the next cycle.
As we discussed, five enzymes took part in the formation of urea. Out of these the first four are found in all cells. But the last enzyme arginase is found only in the liver cells thus assuring the formation of final product urea only in the liver despite the formation of arginine in other tissues.
Energetics of Urea Cycle
Let us take a look at how much energy is consumed during one turn of the urea cycle. On considering only the urea cycle, and not considering the other biopathways linked, to produce one urea molecule 4 ATP molecules are used up as shown below:
NH4+ ions to Carbamoyl phosphate- utilisation of 2ATP
Citrulline to arginosuccintae- breakdown of 1 ATP to AMP + PPi which is equivalent to 2 Pi
Therefore the entire reaction can be summarised as follows:
2NH4+ + HCO3– + H2O + 3ATP + Aspartate → Urea + 2ADP + 4Pi + AMP + 2H+ + Fumarate
Rate Limiting Steps of Urea cycle
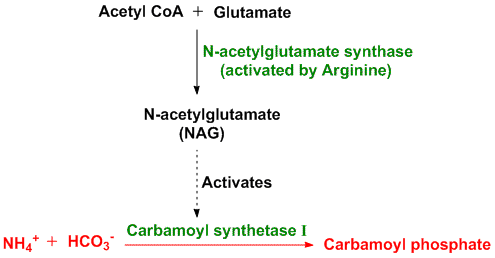
The conversion of ammonium ions to carbamoyl phosphate catalysed by carbamoyl synthetase I is a rate limiting step. This enzyme (carbamoyl synthetase I) gets activated by N-acetylglutamate (NAG) which is formed by reaction between acetyl CoA and glutamate catalysed by the enzyme N-acetylglutamate synthase (activated by arginine).Thus concentrations of glutamate and acetyl CoA as well as levels of arginine determine the steady state levels of N-acetylglutamate (NAG) which in turn regulates the concentration of urea. When a high protein diet is is consumed, levels of NAG increases and in turn urea levels increase. Also during starvation, when muscle proteins start breaking down to source out energy, urea levels increase in response. The rest all enzymes participating in the urea cycle are mostly regulated by the concentrations of their respective substrates.
Recommended Texts
- David L. Nelson and Michael M. Cox, Lehninger Principles of Biochemistry 6th Edition
- Jeremy M. Berg, John L. Tymockzo and Luber Stryer, Biochemistry 7th Edition
- Reginald H. Garrett, Charles M. Grisham, Biochemistry by Reginald H Garrett 5th Edition
.
- U. Satyanarayana, U. Chakrapani, Biochemistry by U. Satyanarayana. 3rd Edition.


thanku soo much for this ….its soo helpful
Hiiii ,
my requested sir
Please,Can you help me
Please suggest some good reference book for 1 st year pharma d students for all subjects pls mam
Very comprehensive can’t be better than this… Thanks a lot
So neat and helpful . Thanks
What is the inhibiting factor for the rate limiting step.if activation if by NAG
Thankyou very much
u r answers r very nice and very usefull to me thanku so much…..