Introduction
Woodward-Fieser rules work well for conjugated dienes and polyenes with upto 4-double bonds or less. Certain plant pigments such as carotenoids have even more than 4 conjugated double bonds. For conjugated polyenes having more than 4 double bonds, the Fieser-Kuhn rules must be applied in order to obtain the wavelength of maximum absorption.
Fieser-Kuhn Rule for Conjugated Polyenes
According to the Fieser-Kuhn rule the following equation can be used to solve for the wavelength of maximum absorption λmax and also maximum absorptivity εmax:
λmax = 114 + 5M + n (48.0 – 1.7 n) – 16.5 Rendo – 10 Rexo
where,
λmax is the wavelength of maximum absorption
M is the number of alkyl substituents / ring residues in the conjugated system
n is the number of conjugated double bonds
Rendo is the number of rings with endocyclic double bonds in the conjugated system
Rexo is the number of rings with exocyclic double bonds in the conjugated system.
and
εmax = (1.74 x 104) n
where,
εmax is the maximum absorptivity
n is the number of conjugated double bonds.
Thus using the above equations, one can get the wavelength of maximum absorbance (λmax) and the maximum absorptivity (εmax)
Sample Problem 1: β-Carotene
β-carotene is a precursor of vitamin A which is a terpenoid derived from several isoprene units. The observed λmax of β-carotene is 452 nm, while the observed εmax is 15.2 x 104. Let us therefore use Fieser-Kuhn rules to calculate the λmax and the εmax for β-carotene.
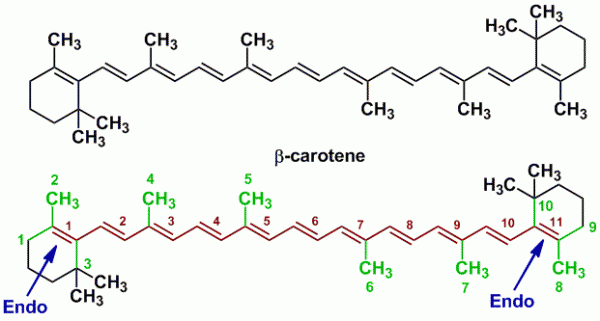
| Name of Compound | β-Carotene |
| Base Value | 114 nm |
| M (number of alkyl substituents) | 10 |
| n (number of conjugated double bonds) | 11 |
| Rendo (number of endocyclic double bonds) | 2 |
| Rexo (number of exocyclic double bonds) | 0 |
| Substituting in equation λmax = 114 + 5M + n (48.0 – 1.7 n) – 16.5 Rendo – 10 Rexo |
= 114 + 5(10) + 11 (48.0-1.7(11)) – 16.5 (2) – 10 (0)= 114 + 50 + 11 (29.3) – 33 – 0= 114 + 50 + 322.3 – 33
Calc. λmax = 453.30 nm |
| λmax observed practically | 452nm |
| Calculate εmax using equation: εmax = (1.74 x 104) n |
= (1.74 x 104) 11Calc. εmax= 19.14 x 104 |
| Practically observed εmax | 15.2 x 104 |
Sample Problem 2: all-trans-Lycophene
Lycophene (all-trans-lycophene) is a bright red carotenoid pigment found in tomatoes and other red fruits and vegetables. However, lycophene has no vitamin A like activity.
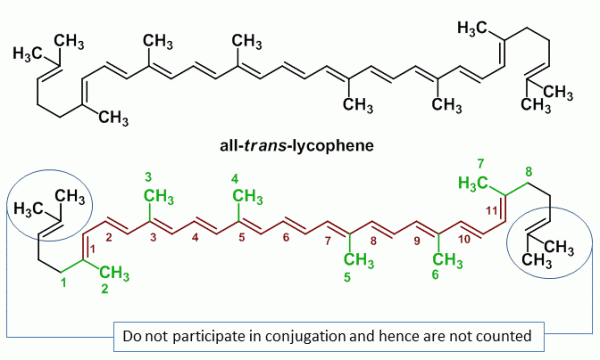
| Name of Compound | all-trans-lycophene |
| Base Value | 114 nm |
| M (number of alkyl substituents) | 8 |
| n (number of conjugated double bonds) | 11 |
| Rendo (number of endocyclic double bonds) | 0 |
| Rexo (number of exocyclic double bonds) | 0 |
| Substituting in equation λmax = 114 + 5M + n (48.0 – 1.7 n) – 16.5 Rendo – 10 Rexo |
= 114 + 5(8) + 11 (48.0-1.7(11)) – 16.5 (0) – 10 (0)= 114 + 40 + 11 (29.3) – 0 – 0= 114 + 40 + 322.3 – 0
Calc. λmax = 476.30 nm |
| λmax observed practically | 474nm |
| Calculate εmax using equation: εmax = (1.74 x 104) n |
= (1.74 x 104) 11Calc. εmax= 19.14 x 104 |
| Practically observed εmax | 18.6 x 104 |
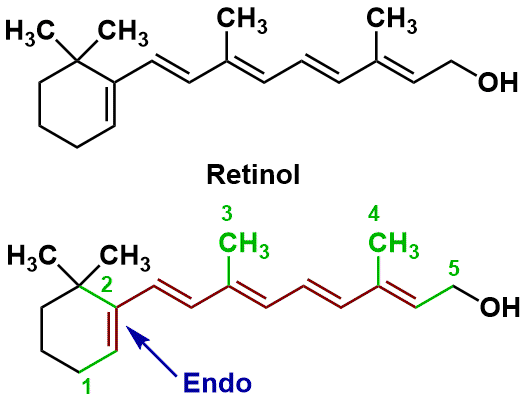
Sample Problem 3: Retinol
Retinol is an animal form of vitamin A and is essential for vision.
| Name of Compound | Retinol |
| Base Value | 114 nm |
| M (number of alkyl substituents) | 5 |
| n (number of conjugated double bonds) | 5 |
| Rendo (number of endocyclic double bonds) | 1 |
| Rexo (number of exocyclic double bonds) | 0 |
| Substituting in equation λmax = 114 + 5M + n (48.0 – 1.7 n) – 16.5 Rendo – 10 Rexo |
= 114 + 5(5) + 5 (48.0-1.7(5)) – 16.5 (1) – 10 (0)
= 114 + 25 + 5 (39.5) – 16.5 – 0 = 114 + 25 + 197.5 – 16.5 – 0 Calc. λmax = 320 nm |
| λmax observed practically | 325 nm |
| Calculate εmax using equation: εmax = (1.74 x 104) n |
= (1.74 x 104) 5Calc. εmax= 8.7 x 104 |
| Practically observed εmax | N/A |
Also Checkout These Other Pages:
- Woodward-Fieser Rules to Calculate Wavelength of Maximum Absorption (Lambda-max) of Conjugated Dienes and Polyenes
- Woodward-Fieser Rules to Calculate Wavelength of Maximum Absorption (Lambda-max) of Conjugated Carbonyl Compounds
- Sample Problems Using Woodward-Fieser Rules
Books on Analytical Chemistry and Spectroscopy
Check out these good books for analytical chemistry and spectroscopy

thanks a lot for providing more detail concerning fieser-kuhn rule
Thank you very much for your work
Be blessed…helps a lot
Good job well done… Please can u add the lambda Max of myrcene and Alfa -tepinene To the examples above.. An my email ID possible… Thanks
thanks,
but i have a question
in retinol,aren’t the alkyl substituent supposed to be four ?
I can’t understand how i consider the ring residue for the cyclic alkene system by using woodward fieser rule.
I calculated Beta- Carotene value as 434 nm. I am happy after seeing above article to know about lambda max value…good job..
Very nice working
nice work…
should have posted video instead…
Thank you! It Helps a lot 😀
Great presentation, and very useful thank you. I was trying to calculate the lamba max for astaxanthin (an analogue of beta carotene). I calculated it to 447.7 nm but i was wondering if the endocyclic carbonyls at the terminal ends of the conjugation would have any alternative effects?
Hi William,
Firstly, the Fieser-Kuhn rules were not developed for carbonyls. Furthermore, in astaxanthin, the carbonyls are actually counted as exocyclic double bonds as they are outside of the ring. Your calculation does come close though to the observed lambda max of 478 nm. However, the rules do not specify anything for carbonyls. If you do find something, please do share it on this page. Thanks.
Great stuff! Can you combine the two equations for retinoic acids?
Thanks Denis. Not sure if we can combine equations for retinoic acids.
Hi ur informations very much helpful to me thanks a lot can u plzzz calculate lembda max of retinol (vitamimA1)
Hi Aaisha,
we have added retinol as sample no. 3 as per your request.
Thank u for sending that rule plz send more ones
Thank you Akul for this nice presentation…