Introduction
A crystal structure is a highly ordered and repeating arrangement of molecules or atoms held together with non-covalent forces. This property of the molecule influences its physicochemical properties.
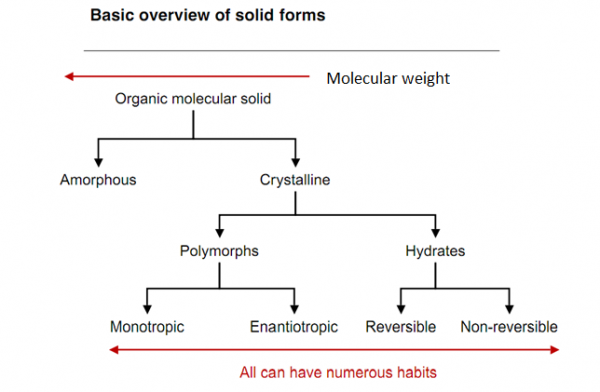
From the above fig 1 we can say that a solid can exist in two forms viz. either amorphous or crystalline. In crystalline form a solid can exist as polymorph, hydrate, solvate, or co-crystal.
Polymorphs arise when molecules of a compound attain different conformations with differing energies of stabilization. Although their chemical properties may be same but they differ in their structural orientation. Generally a less stable polymorph converts into its stable polymorph. But the conversion might take years or few seconds to occur as it depends on the activation energy supplied to the polymorph and free energy of that solid. Lowest energy form is more stable. At transition temperature when one form (polymorph) is more stable than the other forms then they are called as Enantiotropic polymorphs and when the all forms are stable at transition temperature, then they are called Monotropic polymorphs. When the crystal’s repeating arrangement unit has a molecule of solvent attached to it then it is called as a solvate. When the solvate molecule is replaced by water molecule then it is called as hydrate. A co crystal is a distinct solid-state material with unique, unpredictable structure and physical property profile (Christer B. Aakero¨y et al.2008). Different polymorphs give a drastic effect on the dissolution rate which is in turn dependent on the solubility (Tros de Ilarduya et al.1997).
Table 1 (Tros de Ilarduya et al.1997)
From the above table 1 dissolution rates of polymorphic forms of sulindac are compared. It was observed that the solvate form were metastable in nature than the nonsolvate forms.
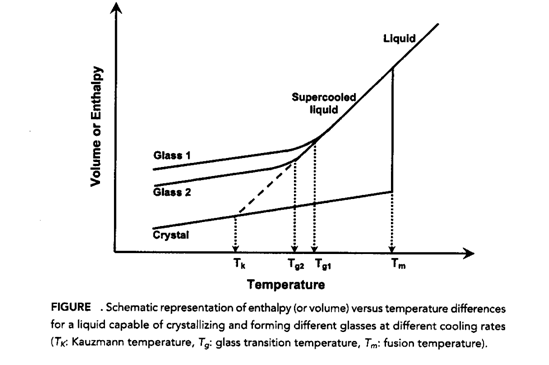
Amorphous state means lack of form. It has no long range order of molecular packing. They generally possess higher internal energy, have greater thermodynamic properties and have increased mobility due to greater intermolecular distances. We can say that an amorphous system is actually fluid but appears to be a solid in the time scale observation. This statement can be explained by the following figure 2:
Figure 2 (Aditya Mohan Kaushal et al.2004)
Cooling from melt, there is a chance that it may convert to a crystalline form at temperature Tm. This is due to an exothermic crystallization which occurs due to decrease in volume and enthalpy. Secondly, if we don’t give enough time for the molecule to crystallize then we don’t see discontinuity at Tm and the system enters into supercooled state. Now on further cooling the viscosity of the compound decreases and it appears to be in a rubbery state. When we further cool it we observe that at some point we don’t see the decrease in viscosity as rubbery state is converted to glassy state. The temperature at which this phenomenon occurs is called as glass transition temperature (Tg). We can say that Tg is a thermodynamic requirement because otherwise the material may get lower enthalpy than crystalline material or even get negative enthalpy Tk. This is a significant difference between amorphous and crystalline materials. Amorphous materials are easy to formulate than crystalline materials but they degrade faster than the latter (generally).
Polymorphism
Acetazolamide (ACZ)
Acetazolamide is used mainly in treatment of glaucoma. It is also used as an antiepileptic and anti diuretic agent. It is formulated as tablets, capsules (for extended release) and injection. It is commercially available as Diamox.
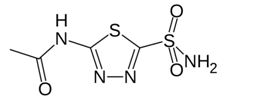
Figure 3: The structure of Acetazolamide (Jenniffer I. Arenas-Garcı´a et al.2010)
The above fig 3 shows the structure of acetazolamide. It has two known polymorphic forms (Form A and B). Form A being the more stable form than form B. Acetazolamide has low solubility and permeability. Hence efforts are being made to form and identify new co- crystals forms of the drug (Jenniffer I. Arenas-Garcı´a et al.2010). Co crystals were made by using four different techniques. Co crystals were prepared by using Solvent drop grinding experiments. Out of 20 Co crystal former used 4-Hydroxybenzoic Acid and Nicotinamide were found to form stable co crystals with acetazolamide. Co crystals formed were characterized using XRPD, IR spectroscopy, DSC and TGA techniques.
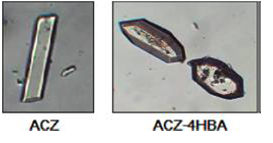
Figure 4: Crystals of acetazolamide (ACZ) and the corresponding co-crystal with 4-hydroxybenzoic acid (ACZ-4HBA) can be distinguished by their different shapes (Jenniffer I. Arenas-Garcı´a et al.2010).
Co crystal phase stability was carried out to check whether the co crystals formed convert to the parent compound ie.ACZ. The co crystals were exposed to water at various physiological pH values. The co crystal formed by ACZ with nicotinamide transformed to ACZ and this was time and pH dependent while co crystal with 4 -hydroxybenzoic acid didn’t transform into ACZ.
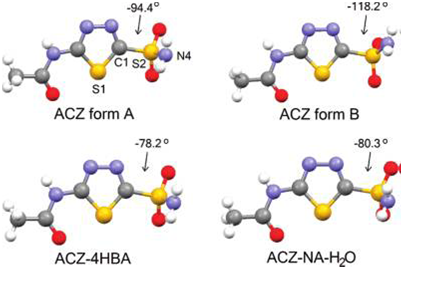
Figure 5: Molecular conformations of ACZ in the crystal structures of ACZ form A,9a,d AC form B,9c ACZ-4HBA, and ACZNA- H2O. N4-S2-C1-S1 torsion angles are indicated (Jenniffer I. Arenas-Garcı´a et al.2010).
Figure 5 gives the molecular conformation of the available polymorphs of ACZ and the two new co crystals formed. These structural details demonstrate that ACZ possesses two potential sites for the formation of doublebridged homo- and heterosynthons, the carboxamidine (C(N)NH) group on the thiadiazole acetamide fragment and the sulfonamide group(Jenniffer I. Arenas-Garcı´a et al.2010).
Thus the co crystals formed were stable enough at ambient temperature. However this study fails to compare the solubility and permeability of co crystals with ACZ.
Paracetamol
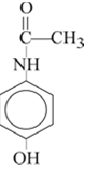
Figure 6 : The Structure of paracetamol (Zimmermann B et al. 2010)
Paracetamol is used as an antipyretic and analgesic. Commercially it is available as tablets, capsules, solutions, suspensions etc. Paracetamol has three known polymorphic form viz. Monoclinic (Form I), Orthorhombic (Form II) and a third instable form III. Efforts were made in characterising form III with use of rapid heating DSC and HPMC (crystal growth modifier) (S. Gaisford. et.al. 2010). Thermal analysis of the polymorphs was carried out using FT-IR spectroscopies (Zimmermann B et al. 2010). Form II is the commercially available form of paracetamol. However form I has a stiffer arrangements in the crystal lattice compared to the orthorhombic metastable form II. Hence form I shows poor compression ability. (Etienne Joiris et.al.1998) Although form II shows better compression ability compared to form I but still form I is preferred as API. This is due to then stability issues related with form II. The stability of the form II varies with alteration in temperature and humidity. Hence, the stability issue prompts the manufacturer to liken form I. Efforts should be made to make form II more stable under ambient temperature and humidity.
Itraconazole
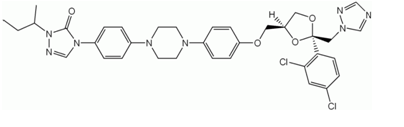
Figure 7: The Structure of Itraconazole (Julius F. Remenar et al.2003)
Itraconazole (Fig 7) is an antimicrobial agent. It is commercially used for treatment of onychomychosis. The main disadvantage for this drug is its solubility. It is a poorly soluble drug. Hence attempts were made to form a co crystal of the drug which will increase the solubility of the drug. Amorphous state of the drug has a greater solubility than the crystalline form but the amorphous form is less stable and degrades quickly in presence of humidity and temperature. Hence in one such attempt a co crystal of itraconazole was prepared using 1,4-dicarboxylic acids (Julius F. Remenar et al.2003). A trimeric co crystal was prepared with itraconazole atoms at the two ends of the dicarboxylic acid (Succinic acid).
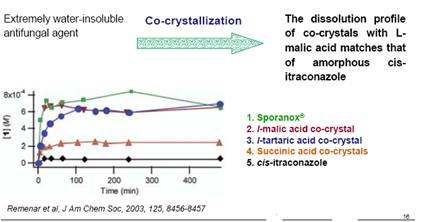
Figure 8: Dissolution profile of itraconazole
From the fig 8 we can conclude that the co crystal with malic acid matches the solubility that of the amorphous itraconazole (Julius F. Remenar et al.2003). Thus this study suggests that it is possible to have a form which matches the bioavailability of the amorphous form but has the physical and chemical stability of the crystalline form.
Fluoxetine
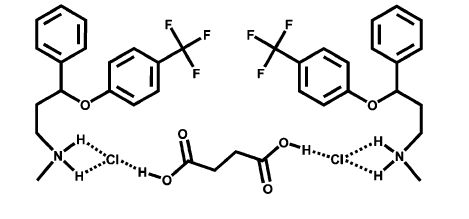
Figure 9: Co crystal of Fluoxetine – Succinic acid (Scott L. Childs et al. 2004)
Fluoxetine is a selective serotonin reuptake inhibitor. It is indicated for acute and maintenance treatment of Major Depressive Disorder (MDD), Obsessive Compulsive Disorder (OCD), and Panic Disorder. (Drugs@FDA). It is commercially marketed as tablets, capsules (for sustained release) and solution (discontinued). Crystalline form of the drug shows poor solubility. Hence efforts were made to develop a co crystal which will have increased solubility compared to the crystalline form. Co crystals were prepared using 1,4-dicarboxylic acids (Scott L. Childs et al. 2004). The above figure 9 shows the co crystal formation between fluoxetine and succinic acid. Co crystallization takes place due to hydrogen bond interactions. The formation of co crystal was confirmed with single crystal structure X-Ray diffraction and IR spectroscopy.
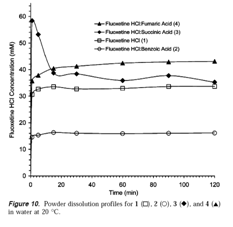
Figure 10: Dissolution profile of the co crystals (Scott L. Childs et al. 2004)
From the fig 10 we can see that distinctive dissolution profiles can be achieved through co crystallization. Hence these forms of API’s can be used to make alternate formulations.
Amorphous Forms
Celecoxib
CELECOXIB is a nonsteroidal anti-inflammatory drug indicated for Rheumatic arthritis. It acts as a COX2 inhibitor. It is commercially marketed as CELEBREX and is supplied in the form of capsules.
Figure 11: The structure of Celecoxib (Patent No. U.S. 6.964,978, 2005)
The crystalline form of the drug is marketed. However it was found that the higher bioavailability was shown by the amorphous state. The downfall of the amorphous state was its stability. This was due to the structural relaxation which causes devitrification of the celecoxib if stored at room temperature (K. Grzybowska et al. 2010). This was enhanced by mixing it with polymers like PVP, PVP-Meglumine which helped in stabilising the amorphous system (Piyush Gupta et al. 2004, Piyush Gupta et al. 2005). A new solid state form was developed by Pharmacia Corporation and patented (Patent No. U.S. 6.964,978, 2005). The polymer used form a composite with the amorphous form and prevents its conversion to the crystalline form. Crystalline form of the Celecoxib is less pharmaceutically active. The amorphous lactose was prepared from the crystalline one by spray drying techniques with isopropanol. Studies were conducted to check the stability and solubility of the drug- polymer amorphous system (Piyush Gupta et al. 2004). The measurement of the crystalline content, glass transition temperatures and enthalpy relaxation were characterised using DSC. Dissolution and solubility studies were carried out to ceck the release mechanisms. Hence by these techniques the new form of Celecoxib was discovered and an improved formulation was developed.
Simvastatin
Figure 12: The structure of Simvastatin (Kirsten A. Graeser et al. 2008)
Simvastatin is a cholesterol reducing agent belonging to the statin family. It belongs to BCS class II (low solubility but high permeability). It has two known polymorphs, however the second polymorph requires extreme conditions to form and form I is the most known stable form available. Simvastatin is suitable candidate for amorphous form. In one such study the physicochemical properties of two differently prepared amorphous forms were prepared and characterized (Kirsten A. Graeser et al. 2008). The two methods were cryo-milling and quench cooling. In quench cooling the compound was melted in oven and then quench cooled by putting liquid nitrogen on the plate carrying the compound. In cryo milling the compound was ball-milled and the mill was surrounded liquid nitrogen. The amorphous material obtained by two different methods were further characterized by using FT-IR, DSC, High Speed DSC, TGA, XRPD, PLS and SEM.
Figure 13: SEM images of simvastatin (a) crystalline, (b) cryo-milled, (c) 4 °C milled, (d) RT milled, (e) recrystallized cryo-milled, and (f) quench cooled.( Kirsten A. Graeser et al. 2008)
Figure 13 depict the difference in the crystal structre of the amorphous material prepared by different techniques. Accelerated Stability studies were carried out on the amorphous material prepared by two techniques. The cryo- milled amorphous product showed less stability compared to quench cooled product. The logic behind making amorphous form is that it shows more solubility than the crystalline form. Amorphous form has more bioavailability compared to the crystalline form. The main disadvantage of this form is that it has excess of thermodynamic properties hence it tends to recrystallize into its crystalline form. If we are able to keep the amorphous form stable enough till the expiry of the medicine then we can achieve a novel and alternative formulation.
Summary
The crystalline and amorphous form of the drug have same molecular configuration but different physical and chemical properties. The crystalline form of the drug is not easy to formulate but the formulator is sure that the formulation will be stable enough till its expiry date. But it is essential to identify the most stable polymorph early in the development. This is of utter importance as the goal is to avoid the scenario of ritonavir where the stable polymorph occurred after manufacturing 240 batches which had impaired bioavailability (Scott L. Childs et al.2004). Compounds used as active pharmaceutical ingredients (APIs) must be screened for polymorphism according to ICH guidelines. Whether to use a hydrate form or a solvate form will depend on the ability of the compound to be stable over varied temperature and humidity. Co crystals offer a new dimension to the formulator as it has the solubility as that of the amorphous material but also retains the physical and chemical properties of the crystalline form. However to choose an ideal co crystal former is of the utmost importance. The amorphous form of the drug has increased solubility than its crystalline counterpart and hence has increased bioavailability. The amorphous material is easy to formulate than the crystalline material due to its high internal energy and increased mobility. But amorphous state is not a state where the molecules love to be and hence the molecules try to give out energy and recrystallize to a more stable crystalline form. This is the major issue with amorphous materials. Little change in temperature or humidity may largely affect the stability of the dosage form. Various pharmaceutical processes affect the final form of the drug. Processes like spray- drying, lyophilisation may cause formation of amorphous form. Amorphous material is formed during mechanical shearing processes (Ball milling, wet granulation, compaction). To conclude one must do extended preformulation studies and then formulate the best possible stable drug available.
References
- Aditya Mohan Kaushal et al.Amorphous Drug Deliver Systems:Molecular Aspects, Design and Performance. Critical Reviews in Therapeutic Drug Carrier System (2004),21(3):133-193.
- Christer B. Aakero¨y et al. Practical Guidelines for Co-Crystal Synthesis.(2008).Pharmaceutical Co-crystals meeting Amsterdam, The Netherlands.
- Etienne Joiris et.al. Compression behaviour of orthorhombic paracetamol. Pharmaceutical Research (1998), Vol. 15, No. 7, 1122-1130.
- Jenniffer I. Arenas-Garcı´a et al.Co-Crystals of Active Pharmaceutical Ingredients – Acetazolamide. Crystal Growth And Design (2010),Vol.10:3732-3742.
- Julius F. Remenar et al.Crystal Engineering of Novel Cocrystals of a Triazole Drug with 1,4-Dicarboxylic Acids.J. Am. Chem. Soc., 2003, 125 (28), pp 8456–8457.
- K. Grzybowska et al. Molecular Dynamics and Physical Stability of Amorphous Anti-Inflammatory Drug: Celecoxib.J. Phys. Chem. B 2010, 114, 12792–12801.
- Kirsten A. Graeser et al. Physicochemical Properties and Stability of Two Differently Prepared Amorphous Forms of Simvastatin.Crystal Growth And Design (2008),Vol.8,No1:128-135.
- Patent No. U.S. 6.964,978.Solid-State Form of Celecoxib Having Enhanced Bioavailability.(2005).
- Piyush Gupta et al. Stability and solubility of Celecoxib-PVP Amorphous Dispersions: A molecular perspective.Pharm Research 2004,Vol 21,No 10, 1762-1769.
- Piyush Gupta et al. Molecular interactions in celecoxib-PVP-Meglumine amorphous system.JPP 2005,57:1-8.
- (-). Prozac. Available: http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseaction=Search.DrugDetails. Last accessed 25th Apr 2011.
- S. Gaisford, et al., Characterisation of paracetamol form III with rapid-heating DSC, J. Pharm. Biomed. Anal. (2010), doi:10.1016/j.jpba.2010.04.017.
- Scott L. Childs et al.A Metastable Polymorph of Metformin Hydrochloride: Isolation and Characterization Using Capillary Crystallization and Thermal Microscopy Techniques.Crystal Growth And Design (2004), Vol.4, No3:441-449.
- Scott L. Childs et al. Crystal Engineering Approach To Forming Cocrystals of Amine Hydrochlorides with Organic Acids. Molecular Complexes of Fluoxetine Hydrochloride with Benzoic,Succinic, and Fumaric Acids.J. Am. Chem. Soc. 126 (2004), pp. 13335–13342.
- Tros de Ilarduya et al. Drug Dev Ind Pharm (1997) 23:1095.
- Zimmermann B et al.Thermal analysis of paracetamol polymorphs by FT-IR spectroscopies.J Pharm Biomed Anal. 2011 Jan 25; 54(2): 295-302. Epub 2010 Sep 21.



1) This article/post is really interesting. Thanks!
2) About paracetamol … maybe it is a typo but i would like to ask to be sure: “Form II is the commercially available form of paracetamol. ” […] “but still form I is preferred as API”. I don’t get it. If Form II is the commercially available form it should also be the one preferred as API (Active Pharmaceutica Ingredient). Am i missing something?