Background Information on Light
It is a well known fact that light is composed of electromagnetic radiation which is often treated as a wave phenomenon which has a defined wavelength. In a wave, wavelength is defined by the distance in a wave where the shape of the wave repeats and is usually determined by the distance between corresponding points of the same phase.
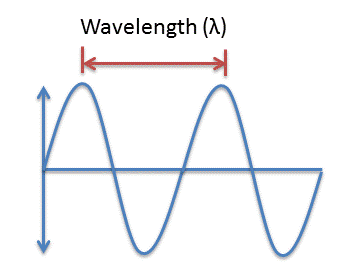
Wavelength being defined as the distance, has the units of distance, i.e. meters (m), centimeters (cm) or even nanometers (nm) and is often denoted by the symbol lambda (λ). The wavelength of light is what determines the photo energy possessed by the light which can be calculated by the equation
E = h.c / λ
Where, E is the energy of the light, h is the Planck’s constant, c is the velocity of light which is a constant and λ is the wavelength. Thus as the wavelength increases, the energy of the light decreases.
Since frequency (ν) = c / λ
The equation may be rewritten as:
E = h. ν
Thus as the frequency increases, the energy of the light increases.
Note – frequency and wavelength are inversely proportional to each other.
The Electromagnetic Spectrum
For a particular type of light the wavelength for it will have a characteristic value which will determine the energy of the light. Visible light has wavelengths ranging from 400 nm (violet) to 780 nm (red). The colors we see with our eyes in our day to day lives have wavelengths between these 400-780 nm. The wavelengths at which different colors are observed is shown in the table below:
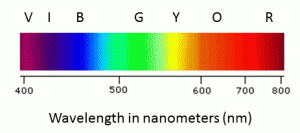
| Color | Wavelength |
| Violet | 400-420 nm |
| Indigo | 420-440 nm |
| Blue | 440-490 nm |
| Green | 490-570 nm |
| Yellow | 570-585 nm |
| Orange | 585-620 nm |
| Red | 620-780 nm |
Note- The order of these colors can be remembered by the mnemonic : VIBGYOR.
White light is a combination of all the colors of light. Thus if you pass white light through a clear glass prism, it will split up to form the different visible colors of light.
To remember the order of the electromagnetic spectrum take a look at the image below along with the text which follows:
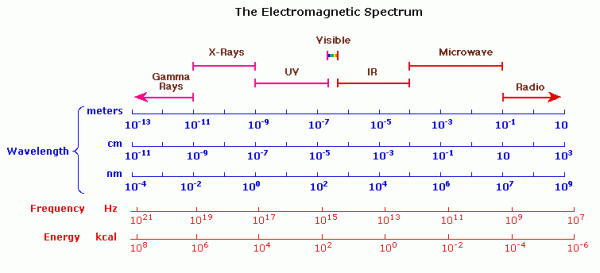
The electromagnetic spectrum is devised by keeping the visible spectrum in the middle from lower to higher wavelengths in the order from left to right. Thus violet will be to the left of the visible spectrum, while red is kept to the right of the visible spectrum.
To the Left:
1] Ultraviolet Spectrum (UV Spectrum) – Moving further to the left of the visible spectrum would lie the ultraviolet region. Although not visible to the naked eye, this region would appear to be violet as it is closer to the violet region of the visible spectrum and hence the name ultraviolet spectrum. It lies between the range of 10 nm to 400 nm.
2] X-Rays – Going to the left of the ultraviolet spectrum, we have first the X-rays (0.01 nm to 10 nm). The X-ray region may also be divided into two regions based on their penetrability:
- Hard X-rays: They are more penetrable, have higher energy and have wavelengths ranging from 0.01 nm to 0.1 nm.
- Soft X-rays: They are less penetrable, have lesser energy and have wavelengths ranging from 0.1 nm to 10 nm.
3] Gamma Rays – Further to the left of X-rays lies the most energetic rays of all, the Gamma rays. These radiations do not have any lower limit of wavelength, but their upper limit lies at 0.01 nm. These rays are of very high energy, and high penetrability.
To the Right
1] Infrared Spectrum (IR Spectrum) – If we move to the right of the visible spectrum, there would lie the infrared spectrum region. Similar to the UV spectrum, the infrared spectrum is invisible to the naked eye, however since the region is closer to the red region of the visible spectrum, it is known as the infrared region. The wavelength range lies between 780nm to 1mm. The infrared spectrum may be split up into further regions:
- Near Infrared (Near IR) – between 780 nm to 2,500 nm.
- Mid Infrared (Mid IR) – between 2,500 nm to 10,000 nm.
- Far Infrared (Far IR) – between 10,000 nm (or 10 μm) to 1000 μm (or 1mm).
2] Microwaves – To the right of the infrared spectrum lies the microwaves. From the name of these waves it can be mistaken that the wavelengths of these waves would most probably be in the micrometer (μm) range. However, they lie in the 1000s of μm range i.e. the range exists in mm. The range for microwaves lies from 1 mm to 10 cm.
3] Radio Spectrum – Finally to the extreme right of the electromagnetic spectrum, i.e. to the right of the microwaves, lies the radio frequency region. This region overlaps at times with the microwave region and can also be said to start at 1mm. However, officially it begins at 10 cm.
Note – for radiations above infrared, i.e. the microwaves and radio frequency, it is common use frequency as a measure of type of electromagnetic radiation instead of wavelength.
Spectroscopy
Spectroscopy is the study of interaction between matter (i.e. molecules, atoms, nuclei) and radiated energy (i.e. electromagnetic radiation). Thus it is the study of how each of the above electromagnetic radiations interacts with molecules, compounds, atoms or even the nucleus. The types of spectroscopy depends therefore on the interaction matter has with radiation:
- Absorption- This is the most common type of spectroscopy. Absorption occurs when the incident electromagnetic radiation is completely absorbed by the matter with which it interacts. Different electromagnetic radiation gets absorbed by different matter species such as atoms and molecules. Absorption is generally measured by monitoring the amount of radiation that is transmitted through the material. Common types of spectroscopy involving absorption are ultraviolet-visible spectroscopy (UV-Vis spectroscopy) or infrared spectroscopy (IR spectroscopy).
- Emission- Emission is the phenomenon when matter itself radiates electromagnetic radiation. The phenomenon of emission may be triggered by an external source of energy such as flames (in case of flame photometry), or even electromagnetic radiation of higher energy (in case of fluorescence). Commonly emission is observed in fluorescence spectroscopy.
- Scattering or Diffraction and Reflection- This type of spectroscopy depends upon how the incident electromagnetic radiation is scattered, diffracted or reflected. An example of scattering or diffraction is X-ray crystallography which employs high energy radiations of x-rays in order to examine the arrangement of atoms in crystals of proteins and molecules.
- Resonance or Coherance- This spectroscopy employs electromagnetic radiation to couple to quantum states of matter in a coherant interaction. An example of this type of spectroscopy is Nuclear Magnetic Resonance spectroscopy (NMR spectroscopy).
- Impedance- It involves the slowing or impeding of the electromagnetic radiation for example, refraction of light.
- Inelastic Scattering- This phenomenon is observed when the wavelength of the incident radiation shifts after it interacts with matter. An example of inelastic scattering is observed in Raman spectroscopy.
The table below gives a list of commonly used spectroscopic techniques used, the type of electromagnetic radiation involved, the type of interaction between the electromagnetic radiation and the matter, as well as the type of matter which can be observed.
| Technique | Type of Electromagnetic Radiation | Type of Matter Observed | Type of Interaction |
| Ultraviolet-Visible Spectroscopy (UV-Vis Spectroscopy) |
Ultraviolet and Visible spectrum | Electrons and electronic excitations | Absorbance |
| Infrared Spectroscopy (IR Spectroscopy) |
Infrared spectrum | Molecular rotations, molecular vibrations, bonds between atoms | Absorbance (or transmittance) |
| Fluorescence Spectroscopy | Ultraviolet and Visible spectrum | Electrons and electronic excitations | Emission |
| Nuclear Magnetic Resonance Spectroscopy (NMR Spectroscopy) | Radiowaves | Nucleus | Resonance or Coherance |
| Flame emission spectroscopy (Flame photometry) | Ultraviolet and Visible spectrum | Atoms | Emission |
| X-Ray Diffraction Crystallography | X-rays | Electron denisities | Diffraction or Scattering |
| Atomic Absorption and Emission Spectroscopy | Ultraviolet and Visible spectrum | Atoms | Absorption or Emission |
Books on Analytical Chemistry and Spectroscopy
Check out these good books for analytical chemistry and spectroscopy
References
- Analytical Chemistry: An Introduction (Saunders Golden Sunburst Series) 7th Ed., by Douglas A. Skoog, Donald M. West, F. James Holler. 1999.
- Fundamentals of Analytical Chemistry 8th Ed., by Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch. 2003.


really helpfull in studies…..
It is a nice article! I have gotten information,that will help me for my work.
thankyou
good article but i need some more information related uv visible light interaction with matter and its effects
I recommend checking out some of the textbooks mentioned above at least for beginning. For advanced you would have to search journal articles for latest information.
Nice article for study. Hope to see some more.