Abstract
This essay outlines brief information about the Human Growth Hormone (hGH). The essay will make you understand the brief idea about the developments taking place in the Pharmaceutical field regarding hGH. The focus of the essay is on the development occurring on the various alternative delivery routes used to deliver the drug to body without use of injections. Hence taking into consideration the patient compliance as well seeing that a good bioavailability is obtained and there is a considerable amount of decrease in dose frequency of the drug. Along with alternative routes, this essay will also provide some information about the newer developments made in the current drugs so that the frequency of dose is reduced. Additional to this new instruments (automated injector with or without needles) are invented so that patient can easily take their daily doses. This essay will also throw some light on the various clinical trials taking place regarding the concerned topic. Many clinical trials which study the other effects caused by the hGH are mentioned. Hence this essay provided a review of critical developments taking place regarding the delivery of hGH.
Introduction
Human Growth Hormone (hGH) also known as somatotrophin is classified as a polypeptide hormone. This hormone is secreted by anterior part of pituitary gland. The main function of hGH is to regulate normal growth in individuals. Abnormalities caused due to hGH are pretty severe as it mostly affects children. Hence interest in curing the disorders caused by this hormone attracted many scientists and vast amount of research has been conducted in finding medicines to cure them. In current market the recombinant somatropin (is a term used for hGH produced by recombinant technology) injections are used for treating disorders related to hGH. (Reference 5.)
Biochemistry
hGH has 191 amino acid long chains connected by disulfide bonds. It is 22kDA in mass. It is non glycosylated.
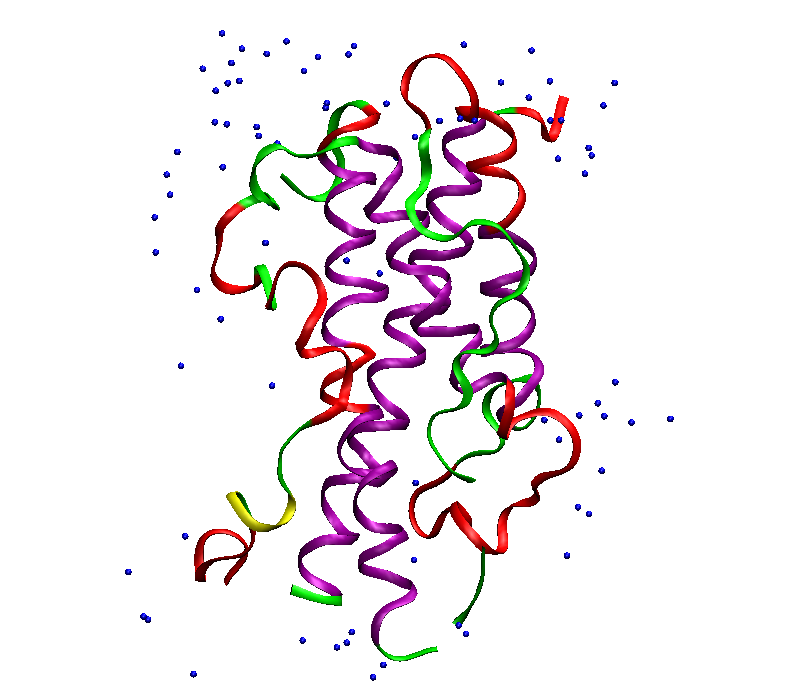
Figure 1 hGH 3D structure
In above figure purple portion indicates the alpha helical structure, while yellow signifies beta helical structure. Blue part shows the presence of water molecules surrounding the hormone. The three dimensional structure consists of 4 antiparallel alpha-helical regions. Helix 4 and 1 has primary sites for binding of the growth hormone receptor (Reference 13.) The expression hGH genes present in the body are of 2 types viz. normal hGH and variant hGH. The variant hGH is found in pregnant women’s placenta and is responsible for growth of the fetus.
Pharmacology
Growth Hormone Secretion and Regulation
(Reference 6)
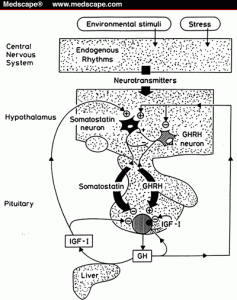
The secretion of GH is controlled by two factors viz.
- Growth hormone releasing factor (GHRH)
- Growth hormone releasing inhibiting hormone (GHRIH) or Somatostatin.
The mechanism works by negative feedback mechanism Due to disruption in the endogenous rhythms the neurotransmitter stimulates the pituitary gland .This causes the release of either of the factors depending upon the need of GH. In case there is need of GH in body then, the pituitary gland causes a decrease in secretion of GHRIH and increase in secretion of GHRH because of which secretion of GH increases. The GH then gets transported to the liver and then it is supplied to the effector organ or cell. The high levels of GH is later are brought back to normal by increase in secretion of GHRIH.
The Growth Hormone Receptor
The hGH receptor is a single chain 620- amino acid trans-membrane polypeptide. It is a member of haemopoietic receptor superfamily (Reference 5)
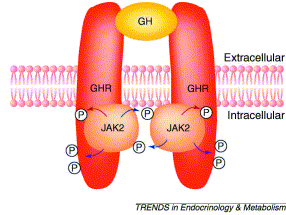
(Reference 9)
Mechanism of action takes place by binding of GH molecule in between the receptor molecules. This binding occurs in the extracellular region and due to this there is receptor dimerization. Due to this there is phosphorylation of the JAK2 protein which causes the desired action.
Pharmacological actions
It regulates growth of cartilages. IGF-1 is responsible for net muscle protein synthesis. It causes elevation of blood glucose level. Hence it is contraindicated with insulin. Its main function is to increase body growth. (Reference 5)
Pharmacokinetics and Metabolism
Recombinant hGH or Pit-hGH is given by Subcutaneous route.This is so because when it was given by IV route it got rapidly cleared from the body and half life was very less and frequent dosing was required to achieve desired effect. The terminal half life of hGH given by IV route was 15-20mins. By IM route there was complete absorption of hGH. But it took a longer time to get absorbed in the system. Due to this there limited availability of hGH in the body and again frequent dosing is required. However this could leave to toxicity and dose dumping. In Sc route there is good bioavailability of the drug. SC administered hGH is 50 to 80 % bioavailable. The drug has longer half life compare to other routes. (Reference 13). By this route the drug may enter the systemic circulation via blood capillaries or lymphatic vessels. The Growth Hormone is metabolised by kidney as well as liver.
Delivery
Recombinant Technology
The official FDA approved hGH are made by recombinant techniques. Earlier hGH was prepared by endogenously removing it from healthy volunteers. But this approached failed as number of patients reported Creutzfeldt-Jacob disease. It is a fatal degenerative neurological disorder. (Reference 5)
This lead to discovery of use of recombinant technology for producing hGH (rhGH).
(Reference 11)
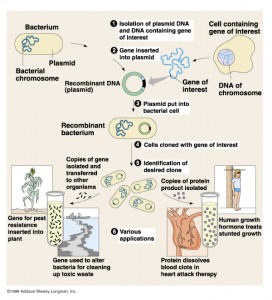
In this process the gene of interest is incorporated in the plasmid. Then it is conjugated to give the recombinant plasmid. It is then cloned and hence the recombinant hGH is made.
Most of the recombinant growth hormones are prepared by incorporating them in bacteria or yeast eg: Humatrope , Neutropin, Genotropin etc…
While some are prepared by using mammalian cells eg: Serostim, Saizen, Zorbtive etc. (Reference 13)
Peptides are highly unstable. Hence to store them is not easy. All these preparations are either lyophilized powder or liquid. Majority of them are in lyophilized form as they are more stable compared to liquid form. The peptide is lyophilized and then it mixed with suitable polymer so that its half life increase and then it is mixed with diluents and packed in single use vials.
Purification of peptides is the main step in preparation. Peptides are contaminated by viruses, host derived proteins, endotoxins, glycosylation variants. Contaminants can be either product related or process related. Purification is required nearly in each step where the product is formed. Such purification techniques should be used which account for less amount of loss of yield. Purification step involves four steps viz.
- Sample Preparation: Here the sample is isolated and purified from the other components by various methods like desalting, buffer exchange etc..
- Capture Step: It is one step ahead than the sample preparation. Here the crude isolated from the other components is purified further to obtain initial product.
- Intermediate Step: After this step we get nearly 60-80% of pure protein
- Polishing Step: Removal of trace elements from the preparation is done. After this process we get nearly 90-100% pure protein. ( Reference 16)
Novel Approaches
Since the approved products are all injection, these are not patient friendly. Hence there is a need to formulate new products which can last for longer period of time in the body and hence reduce the frequency of the dose. Many studies have been carried out to see whether a controlled or sustained release formulation can be made which will drastically reduce dose intervals. In one such study a sustained release formulation of recombinant human growth hormone (SR-rhGH) was developed as a once-a-week injection formulation using sodium hyaluronate. SR-rhGH was produced in the form of solid micro-particles by spray drying technology. Although the study showed to produce a good result but only animal testing was done. Hence human clinical trials are yet to be carried out. (Reference 18).
In another study an ideal sustained release formulation was made of PLGA microspheres using double emulsion method. The results showed a greater residence of activity compared to normal rhGH (Neutropin was used as control). But in vivo testing was carried out in animal models and there was not that significant increase in residence time of dose in the body of animals. (Reference 7)
Alternative approach is by either changing the delivery route of the product or by making some structural or formulation changes in the product. There are many studies going on to check for the alternative delivery routes. Let us look at the various alternative routes and their ability to incorporate rhGH safely and without causing any toxicity.
Alternative Delivery Routes
Oral Route
Generally hGH cannot be given by oral route is because of it poor bioavailability. This is due to gastric enzyme activity on the drug and secondly a large molecule like hGH cannot pass through mucosal barrier. Hence to solve this problem the drug has to be small in size and has to be properly protected. The only logical approach remains is of microspheres.
Microspheres are spherical matrix systems in which drug can be dispersed. These have several advantages over conventional tablets viz:
- Increased residence time
- Increase Stability
- Targeting to site of action etc…
In one such study, Pluronic surfactant was used to stabilize the rhGH during microencapsulation. The drug was incorporated into PLGA microspheres. (Reference 4) However this study was analyzed by using in vitro methods, so we cannot make any solid conclusions from this.
There are also several disadvantages of using PLGA microspheres. So in one study the scientists have used a complex of copoly (dl-lactic/glycolic acid)- zinc oxide. This study further states that by using this complex the release rates of rhGH from microspheres are increased compared to PLGA microspheres. ( Reference 20)
Intranasal Route
This offers the advantage of ease of administration, larger surface area and no first pass metabolism. However the area to which it gets exposed is small hence this can reduce the absorption of the drug. Hence it has to be accompanied with an absorption enhancer. In one such study the intranasal delivery of rhGH was done in sheeps using chitosan based powders. The study showed the in vivo tolerance of chitosan based rhGH. However the results are not pretty accurate and human clinical trials are not conducted. (Reference 19)
Transdermal Route
The main advantage of using this route is that there is no metabolic or chemical degradation of the drug and the drug also dodges the first pass metabolism. However very few amount of drug can get pass through the pores. Hence in the following study the transdermal delivery of rhGH is done through micro channels using radiofrequency. (Reference 3) However for drug to pass through this route an external force is required which neither solves our patient compliance problem nor lessens the frequency of doses.
Other Approaches
Some of the novel approaches involve change in structural property of the drug. For example the residence time of the rhGH was increased by using C-Terminally PEGylated hGH-derivatives. (Reference 2) However large PEG molecules cannot be attached as it will hamper its absorption but a small PEGylated molecule will increase its residence time compared to a non-PEGylated molecule.
Other approach is the preparation of a sustained release product which is done by reaction of the drug with the polymer. One such study uses Dextran T40 which was oxidized with periodate to form aldehyde groups to which recombinant human growth hormone (rhGH) was attached. However human clinical trials are not conducted but the study showed a rapid increase in body growth of the rats. (Reference 10)
Unique approaches
This type of approaches involve in making devices which are needle free and people who have hGH deficiency and take their medicine without visiting the hospital. This approach leads to invention of 2 devices of which one is needle free. These both devices are made by a company named Saizen.
(Reference 17)

Easypod is an injector which has small needle in it attached to a preset dose. This instrument is useful for patients which require continuous and frequent doses of hGH.
Patients which have the fear of needles can use Cool. Click. This instrument is needle free and here also the dose is preset. Hence these 2 inventions solve the problem of Pateint compliance. A clinical trial study shows that use of Cool. Click help in patients with childhood onset of GH deficiency. (Reference 12)
Clinical Use
Officially FDA approved recombinant hGH are clinically used for following:
- Children with short stature caused by GH deficiency, Turner syndrome, idiopathic short stature, SHOX deficiency, and failure to catch up in height after small for gestational age birth.
- Adult Patients: Treatment of adults with either childhood-onset or adult-onset GH deficiency. (Reference 8 )
These above uses are all on label uses, however there are many other off label uses too.
The growth hormone is used for many off label disorders. Many clinical trials are being conducted to study these off label uses.
Some off label uses of hGH are:
- Use of hGH in children with renal disorders: In this the growth of children is affected due to defects in metabolism or defects in IGF-1/hGH axis. This is cured by giving adjuvant therapy of hGH along with the drugs used to cure renal disorder. (Reference 13)
- Certain clinical trials are in process to check whether immunogenicity is caused by administering Saizen injections (Reference 14)
- A study of clinical trial is completed which shows the use of GH helps immune function in patients with HIV. (Reference 15)
- One study is being carried out which is in its Phase 2 of clinical trials. The aim of the study is to study the effect of GH on glucose and protein metabolism in children with GH deficiency. (Reference 1)
There are many other clinical trials being conducted to either study the off label uses of hGH or to study the effects of hGH on other metabolic pathways. Hence a vast amount of research is being carried out in this field but still we are searching for a formulation which decrease dose frequency and would be needle free.
References
- (Aug 2006). Effects of Growth Hormone on Glucose and Protein Metabolism in Children With Growth Hormone Deficiency. Available: http://clinicaltrials.gov/ct2/show/NCT00362063?term=growth+hormone&rank=6. Last accessed 5th Jan 2011.
- Bernd Peschke, a, * Magali Zundel, a Sonja Bak, a Trine R. Clausen, b Niels Blume, c Anja Pedersen, d Florencio Zaragoza a and Kjeld Madsen a. (2007). C-Terminally PEGylated hGH-derivatives. Bioorganic & Medicinal Chemistry. 15 (-), p4382-4395.
- Galit Levin, 1,3 Amikam Gershonowitz, 1 Hagit Sacks, 1 Meir Stern, 1 Amir Sherman, 1 Sergey Rudaev, 1 Inna Zivin, 1 and Moshe Phillip. (April 2005). Transdermal Delivery of Human Growth Hormone Through RF-Microchannels. Pharmaceutical Research. 22 (4), p550-554.
- Gang Wei, Li Fang Lu, Wei Yue Lu. (2007). Stabilization of recombinant human growth hormone against emulsification-induced aggregation by Pluronic surfactants during microencapsulation. International Journal Of Pharmaceutics. 338 (-), p125-132.
- Gary Walsh. (2003). Hormones of therapeutic interest. In:Biopharmaceuticals: Biochemistry and Biotechnology. England: Wiley. p272-279.
- Growth Hormone: Growth Hormone Physiology. Available: www.medscape.com/viewarticle/409613_3. Last accessed 10th Dec 2010.
- H.H. Kwak a,b, W.S. Shim b, M.K. Choi b, M.K. Son a, Y.J. Kim a, H.C. Yang a, T.H. Kim a, G.I. Lee a, B.M. Kim a, S.H. Kang a, C.K. Shim b,. (2009). Development of a sustained-release recombinant human growth hormone formulation. Journal of Controlled Release. 137 (-), P150-165.
- Humatrope. Available: http://www.accessdata.fda.gov/drugsatfda_docs/label/2010/019640s078_2lbl.pdf. Last accessed 18th Dec 2010.
- James Herrington and Christin Carter-Su. (1 Aug 2001). Signaling pathways activated by the growth hormone receptor. Trends in Endocrinology and Metabolism. 12 (6), Pages 252-257.
- John Battersby a’*, Ross Clark a, William Hancock l”a, Estela Puchulu-Campanella b, Neill Haggarty 2,b, Dick Poll b, David Harding. (1996). Sustained release of recombinant human growth hormone from dextran via hydrolysis of an imine bond. Journal of Controlled Release. 42 (-), p143-156.
- Lecture notes for genetics. Available: http://www.google.com/imgres?imgurl=http://www.anselm.edu/homepage/jpitocch/genbio/plasmidtechn.JPG&imgrefurl=http://www.anselm.edu/homepage/jpitocch/genbio/geneticsnot.html&usg=__kfTiuN_yhHhLWo3eg028. Last accessed 15th Dec 2010.
- (May 2005 ). Cool.Click™ Adolescent Transition Study: Study of Saizen® in Subjects With Childhood-onset Growth Hormone Deficiency.Available: http://clinicaltrials.gov/ct2/show/NCT00109733?term=growth+hormone&rank=1. Last accessed 5th Jan 2011.
- Melinda Marian and James Oeswein. (c2008). Growth Hormones. In:Pharmaceutical biotechnology fundamentals and applications. 3rd ed. USA: Informa Healthcare. p281-291.
- (Nov 2010). Saizen® Solution for Injection Immunogenicity Study..Available: http://clinicaltrials.gov/ct2/show/NCT01237340?term=growth+hormone&rank=2. Last accessed 5th Jan 2011.
- (Oct 2003). Growth Hormone to Increase Immune Function in People With HIV. Available: http://clinicaltrials.gov/ct2/show/NCT00071240?term=growth+hormone&rank=4. Last accessed 5th Jan 2011.
- Protein Purification. Available: http://www.bostonbiochem.com/servicepages/protein_purification.php. Last accessed 15th Dec 2010.
- Saizen Delivery Devices. Available: http://www.saizenus.com/deliveryDevices.aspx. Last accessed 6th Jan 2011.
- Sun Jin Kim a , Sei Kwang Hahn a, T, Myung Jin Kim a , Duk Hee Kim b , Young Phil Lee a. (2005). Development of a novel sustained release formulation of recombinant human growth hormone using sodium hyaluronate microparticles. Journal of controlled release . 104, p323-335.
- Yu-Hui Cheng, A. Margaret Dyer, Inderjit Jabbal-Gill, Michael Hinchcliffe, Richard Nankervis, Alan Smith, Peter Watts *. (2005). Intranasal delivery of recombinant human growth hormone (somatropin) in sheep using chitosan-based powder formulations. European Journal of Pharmaceutical Sciences. 26 (-), p9-15.
- Yutaka Yamagata . (2003). Preparation of a copoly (dl-lactic/glycolic acid)-zinc oxide complex and its utilization to microcapsules containing recombinant human growth hormone. International Journal of Pharmaceutics. 251, p133-141.)