Introduction
Approval of health authorities for inhaled insulin was considered as a remarkable step in the history of Diabetes. Pfizer’s dream of bringing another blockbuster product into the market had passed its final stage. In spite of all this, Pfizer failed to encash on its priced asset. The reasons for this will be meticulously retrospected in the following essay. Before we could talk about Exubera, let’s just have a quick look at Diabetes.
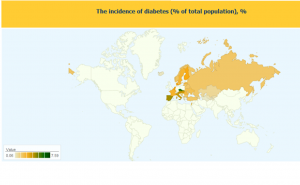
From the above figure, it is clear that diabetes has reached an epidemic status. The only way of curing it is to live on injections of insulin. Insulin and insulin analogues are given as IV or SC injections to the patients. Many other drugs which increase insulin secretion in body are also administered as co therapy with injections. But no one has developed inhaled insulin. Pfizer along with Nektar Therapeutics were the first and till now the only companies to bring inhaled insulin to the market.
Pharmaceutical Aspects of Exubera
Exubera is obtained by recombinant methods. It is grown in E.coli and then isolated. It is a white to off-white powder. It is stored in blisters package. This insulin has empirical formula as C257 H383 N65O77S6 and a molecular weight of 5808 Da. Formulation of exubera is viz:
| No | Name of ingredient | Role |
| 1 | Recombinant Human Insulin | Active ingredient |
| 2 | Sodium Citrate (Dihydrate) | Alkalizing agent |
| 3 | Mannitol | Plasticizer |
| 4 | Glycine | Freeze- dried agent |
| 5 | Sodium Hydroxide | Base |
It enters blood circulation by the alveoli route. Fine particles are capable of reaching to deep lungs. (reference 2). Many clinical trials have been conducted to check the efficacy and safety of Exubera.
In one such trial its release profile was compared with the subcutaneous preparation and normal human insulin. The results found were encouraging. Onset of action of Exubera was slightly faster than SC preparation and Human Insulin (reference 3)
Not much of the difference was found in efficacy and safety of Exubera and SC preparations (reference 3, 4). The main advantage of Exubera over SC injections is that, its absorption is not dependent on body mass index of the patient. But before administering Exubera the physician needs to check the patient’s ability to breath in and accordingly the prescription was given.
Device
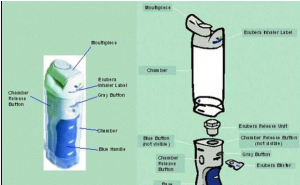
Extend the base of the inhaler and put the blister pack in the gap. Press the blue handle to create pressure in the inhaler. This will cause the release of the insulin. Once you see the white cloud in the transparent chamber then open the mouthpiece and inhale the insulin. The blister packs comes in 2 doses viz : 1 mg (green) and 3 mg (blue). Clean it with soft wet cloth after taking the dose.
Reasons For Failing
Although Pfizer became successful in launching a novel form of insulin, yet it still couldn’t recover its target sales. This inability of Pfizer to capture the market inspite of patenting the product raised many eyebrows. This blockbuster drug proved a disastrous strategy for Pfizer for the following results:
- Contraindication: This was not accounted as the main reason for failing of the drug but it contributed to its decrease in sales. Exubera was contraindicated in smokers. In smokers it showed variable absorption and hypoglycemia. There was increase in onset of action. It is also contraindicated in people who are heavy passive smokers. Studies were still carried out to prove this data. Therefore, Exubera could not be prescribed to those patients who either smoked or are under constant passive smoking.
- Other studies: Extensive studies were yet to be carried out to show effects in pregnant women, people with liver toxicity and renal toxicity. Hence people with these diseases and pregnant women were skeptical in using Exubera. In one such study revealed that, during clinical trials 6 volunteers were diagnosed with cancer. This created fear in the minds of the people.
- Ease of using the instrument: The instrument as you can see was not user friendly. The bulkiness of the instrument was not appealing to the patient (reference 1). People were discouraged by seeing the complexity of the inhaler compared to inhalers used for asthma.
- Economic Reasons: The major setback this product got was from NICE (health authorities) in UK. It challenged the reason of Pfizer which states that it will be a substitute for those who have needle- phobia. By using Exubera the cost of NHS was going to increase to £1,102 annual cost from £300 a year (for SC injection) (reference 5). NICE argued that a very small number of people suffer from chronic needle-phobia and Exubera is not a sole alternative to it as these patients still have to prick themselves for blood tests to check glucose level and also for taking SC or IV injections of long-acting insulin. NICE said that it could not justify the use of Exubera and the cost increase due to it.
Conclusion
In the year 2007, one year after launching the product Pfizer decided to withdraw the product from the market. The official reason for the withdrawal was given as poor sales but the underlying reasons for its failure to gather sales were not mentioned. Pfizer still holds the patent for Exubera and was facing legal action of patent infringement from Novo Nordisk. The case got settled later on (exubera-risks.com). The blockbuster drug became a disaster for Pzifer because of its negligence to conduct a market research and look at the needs of consumer and preparing the design accordingly. Due to this, around 660 employees lost their jobs and Pfizer had a loss of $2.8 billion (reference 1, 7). In the end, we must agree that although Exubera was great invention but it lacked the potential to be a blockbuster drug.
References
- Arlene Weintraub. (2007). Pfizer’s Exubera Flop. Available: http://www.businessweek.com/technology/content/oct2007/tc20071018_028695.htm. Last accessed 21 Mar 2011.
- FDA Data (2006). Exubera. Available: http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/021868s016s017lbl.pdf label. Last accessed 21 Mar 2011.
- Klaus Rave et. al. (2005). Time-Action Profile of Inhaled Insulin in Comparison With Subcutaneously Injected Insulin Lispro and Regular Human Insulin. Diabetes Care. 28, p1077-1082.
- M.C. Liu, R.J. Riese, K. Van Gundy, P. Norwood, B.E. Sullivan#, P.F. Schwartz and J.G. Teeter. (2008). Effects of inhaled human insulin on airway lining fluid composition in adults with diabetes. European Respiratory Journal. 32 , p180-188.
- Sarah Hall. (2006). Health watchdog refuses to approve insulin inhaler. The Guardian. Available: http://www.guardian.co.uk/society/2006/apr/19/health.medicineandhealth?INTCMP=SRCH. Last accessed 12 Mar 2011.
- The incidence of diabetes (% of total population). Available: http://statinfo.biz/Geomap.aspx?region=world&act=8154&lang=2. Last accessed 21st Mar 2011.
- Exubera Inhaled Insulin: Reported & Suspected Risks for Diabetics. (2008). Available: http://www.exubera-risks.com/. Last accessed 21 Mar 2011.