Introduction
The skin is the most superficial part of the body. The signs of ageing are most visible in the skin. Although, ageing skin is not a threat to a person, it can have a detrimental effect on the psychology of a person. A look into the causes of skin ageing, the available treatments and preventive measures for this inevitable change is important to help both the already aged, as well as, the youth.
This is a 4 part article in which:
- Part 1 – Discusses the structure of skin and its different components
- Part 2 – Discusses cutaneous ageing and its causes
- Part 3 – Discusses the characteristics of ageing skin and the changes in skin appearance
- Part 4 – Discusses products and treatments for skin ageing
a) Sunscreen Agents
b) Moisturizers
c) Antioxidants
d) Make Up
e) Dermal Fillers (current article)
f) Chemical Peels
g) Botulinum Toxin
h) Estrogen and Hormonal Treatments
i) Plastic Surgery
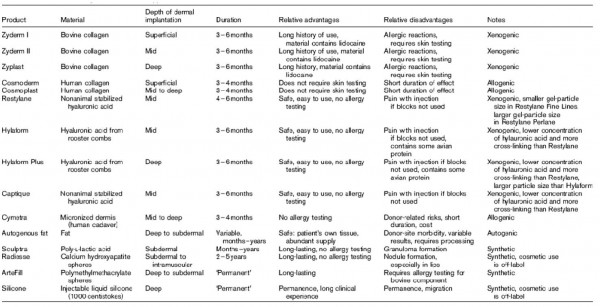
Dermal Fillers1,2
Dermal fillers have expanded and enhanced non‐surgical option in facial rejuvenation. Age changes in face include alteration in skin quality, volume depletion of soft tissue and bones and gravitational descent of facial muscle. The cumulative results are fine lines, wrinkles, folds and furrows. Facial fillers are designed to address rhytids (folds and furrows) and soft tissue loss. It can be used anywhere, especially useful in the lower third of the face. Injectable fillers maybe used in forehead, glabella, nasal tip, eyelids, cheeks, nasolabial folds, melolabial folds and lips. Filling agents use their volume occupying properties to replace lost subdermal fat and dermal proteins.
COLLAGEN
Collagen lays the structural foundation of the skin. With age the amount of collagen in the skin decreases. Collagen dermal fillers were developed as logical replacement for dermal constituent lost thru ageing.
Bovine Collagen:
They are the original non autologous injectable fillers. This collagen is derived from cow hides. There are three types of bovine collagen available in the market: Zyderm I, Zyderm II and Zyplast. Zyderm I composes of 3.5% collagen packed in phosphate buffer solution along with 0.3% lidocaine. It is usually injected in superficial papillary dermis. Zyderm II consists of 6.5% collagen by weight. It is injected in the mid dermis region of the skin.
Zyplast provides longer duration of correction as in this collagen fibrils are cross linked with glutaraldehyde, thus, inhibiting degradation by collagenase. Zyplast is generally injected in deep dermis. Lidocaine in solutions improves comfort of injection and also inhibits activation of eosinophils that play a role in bruising and edema. Collagen’s platelet aggregating effect may also inhibit bruise formation. Allergy testing prior to administration of these products is required via small skin inoculation as some patients are known to develop hypersensitivity to bovine collagen following repeated injections. Many recommend a second test a few weeks after the first, as the first test would act as the sensitizing dose.
Zyderm I requires over correction by 100% to compensate the loss of saline from injected material. With Zyderm II 50% over correction is recommended. While no over correction is required in case of Zyplast fillers. As the saline is absorbed a network of collagen forms to restore skin contour. After a period of a month, host connective tissue grow into the network giving it texture and appearance of normal host tissue. The injected collagen is eventually detected as foreign substances and degraded by collagenase and inflammatory cells. Eventually the foreign body reaction clears the material within a duration of less than 6 months.
Human Collagen:
The FDA approved human collagen are Cosmoderm and Cosmoplast. They are used for the restoration of lip border, correction of facial wrinkles, acne scars and other soft tissue contour deformities. These products contain purified collagen from human fibroblast cell culture. Processing renders the product free from immunological cells and melanocytes. It obviates need to allergy testing.
Zyderm, Zyplast, Cosmoderm, Cosmoplast are packed in syringes containing 0.3% lidocaine as local anesthesia. Cosmoplast, like Zyplast, contains collagen fibrils linked with glutaraldehyde for increased duration of action. Human collagen is easier to use than bovine collagen, with smoother injection and less lump formation. The advantage of injectable collagen cultured from the patient’s own skin may represent an attractive but expensive alternative.
HYALURONIC ACID
It is a major component of the connective tissue matrix in the dermis. Unlike collagen its chemical structure is uniform throughout living species thereby decreasing its immunogenicity. Older skin has lower levels of Hyaluronic acid with resultant loss of tissue hydration. Hence, use of Hyaluronic acid fillers seems ideal, but lasts only 1‐2 days in natural form, as it is locally degraded and eventually metabolized to carbon dioxide and water in liver.
Thus to increase the longevity of Hyaluronic acid fillers, they are stabilized by cross linking process. Cross linked hyaluronan is called hylan. Hylans or hyaluronic acid gels are swollen with water and have unique attribute of dynamic viscosity. Under pressure of injection the gel can pass through a small gauge needle. On removal of the shearing force, viscosity increases and thick gel, that is unlikely to migrate develops at site of tissue implantation. Unique characteristic of hyaluronic acid fillers is isovolemic degradation.
Concentration of gel decreases during reabsorption, but volume remains same until last molecule of hyaluronic acid are degraded. Such an implant maintains 95% of initial space filling volume till last of material is completely reabsorbed. It is approved for mid to deep dermal implantations for correction of moderate to severe facial wrinkles, effective for lip shaping and augmentation. No allergy testing is required for these products.
There are four hyaluronic acid preparations approved by the FDA.
1] Restylane is FDA approved. Restylane is produced from cultures of Streptococcus equi. The resulting hyaluronic acid chains are chemically stabilized through permanent cross‐linking with epoxides. Restylane is 1%crosslinked. During processing, the hyaluronic acid concentration increases approximately 4‐ fold to 20mg/ml and the small gel‐particle size is formed (400mm). Low levels of impurities, high concentration of hyaluronic acid, and minimal cross‐linking help increase the biocompatibility of Restylane with native hyaluronic acid. This helps achieve maximum implant residence time. Other preparations of Restylane that are not yet FDA‐approved are Restylane Fine Lines (indicated for superficial lines and more superficial implantation) and Restylane Perlane (indicated for volume contouring and deeper implantation). Each has identical hyaluronic acid concentrations (20 mg/ml), but differs in the size of the particles. Viscosity is proportional to the size of the particles (Restylane Perlane has the largest particles, Restylane Fine Lines has the smallest particles).
2] Hylaform (INAMED Aesthetics) is FDA approved. It uses hyaluronic acid derived from the dermis of rooster combs that is chemically cross‐linked with divinyl sulfone. It does contain a small amount of avian protein. The concentration of hyaluronic acid is 5.5 mg/ml. The total cross‐linking of hyaluronic acid in Hylaform is 20%. The greater degree of cross‐linking in Hylaform may reduce biocompatibility within the dermis and, along with a decreased concentration of hyaluronic acid compared to Restylane, result in decreased duration of the cosmetic effect. Hylaform Plus (INAMED Aesthetics) is similar to Hylaform except for its larger particle size (750 compared with 500mm) and is intended for deeper dermal implantation.
3] Captique (INAMED Aesthetics), like Restylane, is non‐animal stabilized hyaluronic acid dermal filler produced by bacterial fermentation. Like the other INAMED hyaluronic acid products, the concentration of hyaluronic acid is 5.5 mg/ml with 20% cross‐linking.
4] INAMED Aesthetics has submitted a premarket approval application to the FDA for a new line of hyaluronic acid fillers called Juvederm. This bacterially derived product differs from the other hyaluronicacid products in that it is a homogeneous gel, rather than gel particles, with the potential for increased biocompatibility and duration.
Each of the hyaluronic acid preparations is injected intradermally. If injected too deep or intramuscularly, the duration of effect may be reduced due to product absorption. If injected too superficially, there may be visible areas of excess fullness and/or skin discoloration. Treatment recommendations are to inject to 100% of the desired volume with no need to overcorrect. Hyaluronic acid as compared to collagen do not contain local anesthesia.
Complications of using hyaluronic acid are
- Increased viscosity compared to collagen result in more discomfort during injection.
- Increased bruising may be noted as structure of hyaluronic acid is similar to heparin.
- Excess bruising will decrease duration of action of product by inflammatory mediators and generating free radicals. The treatment may last up to 9 months.
BIOLOGICAL COMBINATION FILLERS
Cymetra is micronized form of Alloderm. Alloderm is an acellular dermal matrix derived from donated human skin tissue. It acts as a framework to allow in growth of native tissues. In addition to collagen it also contains dermal elements, elastins and proteoglycans. They are preserved as freeze dried particles which can be reconstituted before use in 1ml lidocaine or saline. Host fibroblast and collagen infiltrate the implanted material. It lasts for 4‐6 months. And skin testing is not necessary. As its effect is dependent on the host’s ability to elicit fibroblast ingrowth and neovascularization, the results can be variable.
AUTOGENOUS FAT
Is the original dermal filler material. It can be harvested from abdomen, thigh and buttocks. Subdermal injection of fat cells in the face has advantage of using self tissue and potential of achieving permanent results (7 years or more). Disadvantage of this filler is that there is unpredictable degree of localized reabsorption with resulting unpredictable cosmetic effect.
POLY-L-LACTIC ACID
It is a FDA approved filler. Sculptra is injectable poly‐L‐lactic acid, a synthetic polymer of the alpha hydroxy acid family. Allergy testing is not required. Microparticles of poly‐L‐lactic acid is 40‐63 μm in diameter and suspended in sodium carboxy methyl cellulose gel. Particle size is large enough to avoid phagocytosis by dermal macrophage or passage through capillary, but small enough to be injected.
It is supplied as powder that can be reconstituted with 3‐5ml sterile water for injection. It should be reconstituted for at least 2 hours in advance for full hydration. Lidocaine may be added to decrease pain of injection. It is injected in subcutaneous tissue or in deeper parts of dermis. After implantation poly‐L‐lactic acid degrade and ultimately gets metabolized to carbon dioxide and water.
Collagen production in response to implant provides long term volume correction. Because of the mechanism, it is avoided to give over correction. Post injection massage are recommended. Sculptra should probably be thought as a diffuse volume enhancement rather than a wrinkle filler. Sculptra is not recommended for lip augmentation or very superficial fine lines. Duration is 18‐24 months.
RADIESSE
It is used for correction of facial lipoatrophy and nasolabial folds. Its primary component is calcium hydroxy apatite spheres, ranging from 25‐45 μm in diameter, suspended in an aqueous gel with glycerin and sodium hydroxy cellulose. Hydroxy apatite is mineral component of bone, so its biocompatibility is excellent and no allergy testing is required. The product is highly viscous but can be injected with 27 gauge needle. Subdermal or intramuscular placement is recommended because of its viscosity. Massage after injection is very important.
Treated areas resemble native soft tissue due to collagen in growth. The carrier vehicle is gradually absorbed, degraded and undergoes phagocytosis. Fibrous encapsulation of particles takes place, which limits their migration. With time the hydroxyl apatite particles will degrade to calcium and phosphate ions. Implant material persists for at least 6 years with no evidence of bone formation. Radiesse is used for deeper folds, furrows and creases. It is more effective in reducing facial wrinkles in nasolabial and melolabial folds than collagen fillers. It is avoided in the lip.
ARTEFILL
Polymethylmethacrylate microspheres are used for facial volume augmentation. Artefill composed of polymethylmethacrylate microspheres suspended in 0.3% lidocaine, 3.5% bovine collagen, 0.3% NaCl, and phosphate buffer pH 7.3. Microspheres comprise of 20% of the mixture and 80% collagen. Range of sphere is from 30 to 40 μm. It is small enough to allow subdermal placement using 26 gauge needle, but large enough to prevent phagocytosis. Allergy testing is required. Collagen is rapidly broken down by collagenase and phagocytized by macrophages with 1‐4 months. During the same period, microspheres become encapsulated with collagen fibers by body’s own fibroblasts. Artefill is placed within deep dermis or sub dermis and is not designed for treatment of fine wrinkles. Initially degradation of collagen vehicle takes place. 50‐75% of the product left behind is maintained because of polymethylmethacrylate.
Polymethylmethacrylate microspheres remain permanently. Accordingly, patients should be advised that complete correction will require two to four treatment sessions, at intervals of 3–4months. Overcorrection at the initial treatment session should be avoided as the body may respond better by being able to encapsulate a smaller quantity of product more frequently. Patients should be advised to minimize facial expressions for 3 days after treatment to reduce likelihood of muscular contractions pushing the substance more deeply into the subcutaneous tissue.
As with nonbiologic fillers, the success of ArteFill depends on the amount of connective‐tissue reaction that mounts in response to the microspheres. Younger individuals have a greater response than do the elderly. The polymethylmethacrylate microspheres remain permanently. Results in some patients have endured 10 years.
INJECTABLE LIQUID SILICONE
A silicone is a polymer synthesized based on the element silicon. Silicon is the 7th most common element in the universe and the 2nd most common in the earth’s crust. It can exist as solids, gels, foams, liquids depending on degree of polymerization. Silicone is inert, clear, and oily, liquid derived from silica and composed of polymerized dimethylsiloxane. Silikon and Adatosil are two products found commercially. They differ in their viscosity such that Silikon (1000 centistokes) is more preferred as compared to the higher viscosity Adatosil (5000 centistokes). It is injected using a micro droplet technique.
The formulation flows easily through the 27‐30 gauge needles. Minute volumes are injected at multiple sites into deep dermis. A grid of injections spaced at 1‐3mm is created in the area requiring augmentation. Over correction with this filler should be avoided. The injected silicone disperses rapidly and becomes individually encapsulated by fibrous tissue. It is used for volume augmentation in lips and is effective in reducing fine lines. It is the most permanent and least antigenic filler.
TABLE SUMMARIZING DERMAL FILLERS
COMPLICATIONS OF DERMAL FILLERS
No procedure is free from complications and dermal fillers are no exception. A myriad of complications have been reported in detail. Some issues related to potential complications are discussed elsewhere in this paper. The technique of injection can create complications if filler material is inadvertently administered at the improper skin depth (skin changes, excessive lumps), at the improper location (product misplacement), or in the improper volume (palpable lumps, contour deformity).
Acute hypersensitivity reactions can occur with any substance but remain a serious concern for materials containing bovine or other animal products. Potential transmission of bovine spongiform encephalopathy (BSE) remains a risk, but is difficult to quantify.
All fillers create some form of histological soft‐tissue reaction that evolves over time. Granuloma formation can occur with any substance that is injected. Granulomatous tissue reactions have been reported even with biologic products, including hyaluronic acid preparations. These granulomas can be treated conventionally with incision/drainage or excision. Hyaluronic acid reactions can be treated uniquely with hyaluronidase. Fillers containing alloplastic materials seem more likely to develop histopathologic foreign‐body reactions with granuloma formation. These potential complications are unique when compared with traditional surgery and warrant unique informed consent of the patient receiving filler agents.
References
- Elderly skin and its rejuvenation: products and procedures for the aging skin, Marcia Ramos‐e‐Silva et. al., Journal of Cosmetic Dermatology, 6, 40–50
- Dermal filler agents: a practical review, Sukhjit S. Johl and Richard A. Burgett, Current Opinion in Ophthalmology 2006, 17:471–479

Thank you for this review and for your website on the whole. I’ve just shared it.