Introduction:
Ginger is a herbal drug used in medication as well a culinary spice and its medicinal use dates as back as to ancient China and India. The Chinese pharmacopoeias, the Sesruta scriptures of Ayurvedic medicine as well as Sanskrit writings give references to the use of Ginger. It is commonly also known as ginger root, zingiberis rhizoma or black ginger. Ginger is in the official pharmacopoeias of several countries including Austria, China, Egypt, India, Great Britain, Japan, Switzerland and the Netherlands. Ginger is also approved as a dietary supplement in the USA and as a non prescription drug in Germany. Ginger and its constituents have numerous medicinal properties such as anti-emetic, carminative, stimulant, anti-inflammatory, cardiotonic, antioxidant, antitussive, antibacterial, cholagogue actions as well as to promote gastric secretions, increase intestinal peristalsis, lower cholesterol levels and anti-thrombotic.
Ginger
| Biological Source |
Dried rhizome of Zingiber officinale |
| Family |
Zingiberaceae |
| Geographical Source |
Indigenous to tropical areas such as Asia (India and China),West Africa, Jamaica. |
| Morphology/Macroscopical Characters |
- The perennial plant is 2-4 feet tall with a green purple flower in terminal spikes.
- The rhizome of ginger is buff colored with longitudinal striations or is fibrous with no cork. The rhizomes are laterally compressed with a length of around 2.75 to 6 inches and 1 to 1.5 cm width.
- The branches (1-3cm length) arising from the rhizome are oblique to it and end in undeveloped buds or depressed scars.
- Ginger can be either ‘coated’ or ‘unscraped’ depending on the amount of cork removed.
- When the rhizome is broken it yields a short fracture with fibrovascular bundle fibers protruding from the broken surface.
- The rhizome is associated with an agreeable aromatic odor and pungent taste, owing to it’s use as a spice.
|
| Microscopic Characters |
- The transverse section of the rhizome shows a zone of cork tissue which is differentiated on the arrangement of cells. The outer zone of cortical cells in the cork are suberized (deposited with suberin which can be stained with fluoral yellow) without division and hence are irregularly arranged.
- The inner zone contains cortical cells which arrange in a radial row and produced by tangential division. The cork cambium is not differentiated.
- Inner to the cork cells is the cortex composed of cortical cells. The cortical cells contain plenty of simple, ovoid (sack-shaped) starch grains (sized 5-60μm) which can be stained with iodine. Inner to the cork is a broad cortex.
- The outer cortex is composed of mainly flattened parenchyma, while the inner zone is composed of mainly normal parenchyma. The cortex also holds suberized oil cells which hold a yellow-brown oleoresin.
- The inner cortex contains generally three layers of closed, collateral vascular bundles which contain phloem. The larger vascular bundles are protected in a sheath of nonlignified fibres. The vascular bundles contain sieve tubes and a xylem with reticulate thickened vessels (these do not stain with phloroglucinol and HCl) which are some times accompanied by secretion cells (holding dark contents).
- Inner to the cortex lies a single layer of endodermis which is devoid of starch.
- Going inwards further from the endodermis lies the outermost layer of the stele which is characterized by a single-layered pericycle. The vascular bundles of the stele resemble that of the cortex with the exception of a ring of small scattered bundles within the pericycle. The stele is mainly composed of parenchyma containing starch and oil cells similar to the cortical parenchyma. The innermost vascular bundles of the stele may contain a fibrous sheath.
|
| Chemistry |
An oily liquid comprising of homomlogous phenols called gingerol (cardiotonic compounds) accounts for the pungency as well as the pharmcological activity of ginger. Gingerols upon dehydration form shogaols and further degrade to zingerone.
![[6]-Gingerol [6]-Gingerol](https://pharmaxchange.info/wp-content/uploads/2013/01/gingerol.gif)
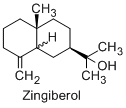
Apart from the gingerols ginger constitutes of 50-70% carbohydrates present as starch, 3-8% lipids which includes free fatty acids like linoleic, linolenic, palmitic, oleic, capric, lauric and myristic, triglycerides and lecithins. The resinous matter constitutes 5-8% and volatile oil around 1-2% of the constituents in ginger. The aroma of ginger is mainly due to oil of ginger which contains a mixture of over 50 constituents. Volatile oils consist mainly of the sesiquiterpenes (β-besabolene and zingiberene), monoterpenes (β-phelandrene, cineol, borneol, citral) and the sesquiterpene alcohol zingiberol.
|
| Chemical Tests |
Suberin can be stained with fluoral yellow and checked with fluorescence. Starch can be stained by iodine and checked under the microscope.
To identify adulterants of ginger in the form of capsicum and grains of paradise, the tincture of the ginger sample may be heated with caustic alkali at 90-100 °C. The liquid is then evaporated, dissolved in dilute HCl and extracted with ether. The ethereal layer is evaporated and the residue which remains should not have be pungent. This indicates that gingerol has decomposed in the alkali solution. Capsaicin and paradol are not that susceptible to alkaline decomposition and would therefore still give a pungent odor. |
| Adulterants/Allied drugs/ Substitutes |
Allied drugs:
Japanese ginger derived from Z.Mioga contains the volatile oil which gives the drug a bergamot like odour and the taste is less pungent than the Z.officinale. Also it differs in physical properties and the starch grains are compound and less eccentric compared to Z.officinale.
Galangal rhizome derived from Alpinia officinarum.
Adulterants:
Being an underground root ginger may be contaminated with worms and attention must be paid for presence of insect fragments. Also ginger can be adulterated by other vegetable adulterants. This can be detected through microscopical examination. Ginger can also be adulterated with ‘spent ginger’ which is ginger from which essence has been extracted, and hence would yield lower than official standards for extracts. Ginger is often contaminated with capsicum or grains of paradise in order to increase pungency. |
| Uses |
- Ginger is used as an anti-emetic and it has been proposed that the aromatic, carminative and possibly absorbent properties of ginger improve the effects of motion sickness directly in the GI tract unlike the anithistamines. It also is effective in the control of vomiting and nausea in hyperemesis gravidarum as well as postoperative nausea and vomiting.
- The cardiovascular activity shown by ginger is mainly due to the gingerols and shogaols. The proposed mechanism for these activities is due to the inhibition of prostaglandin synthetase thus hampering prostaglandin biosynthesis by the gingerols.
- Ginger also has antiplatelet aggregation property which is due to the inhibition of thromboxane synthesis.
- Ginger also lowers cholesterol levels by inhibition of cholesterol biosynthesis under the assumption of inhibiting HMGCoA reductase.
- Ginger also possess antitussive, anti pyretic and analgesic effects.
- Ginger rhizome also has been reported to have fungicidal, antibacterial and anthelmintic properties.
- Sesquiterpene hydrocarbons are responsible for the anti- ulcer activity.
|
| Other Notes (life cycle, extraction etc.) |
|
| Adverse Reactions |
Ginger in high doses can cause CNS depression or cause hindrance with cardiovascular activity. |
References:
- Trease and Evans’ Pharmacognosy, 16e (Evans, Trease and Evans Pharmacognosy)
Elsevier: New York, 2009.
- Ara DerMarderosian, et. al. The Review of Natural Products 4th Edition.
- Kokate, C. K.; Gokhale, S. B.; Purohit, A. P. A textbook of Pharmacognosy, 29th ed.; Nirali Prakashan: Pune, 2009.

![[6]-Gingerol [6]-Gingerol](https://pharmaxchange.info/wp-content/uploads/2013/01/gingerol.gif)

there is no structure of microscopy of zinger drug