Aldol synthesis gives rise to stereocenters in the molecule. Directed aldol synthesis is an approach in order to control the stereochemistry of the product being formed. Part 2 considers how to control whether syn products or whether anti products are formed utilizing the Zimmerman Traxler principle of stereocontrol.
Part 1 has discussed already the formation of Z-enolate and E-enolate. Once we have a established a method for selective formation of enolate, aldol reactions are then selective for their corresponding enolates. That is to say, Z-enolate will produce syn product, while E-enolate will produce an anti product. This is controlled due to formation of another cyclic intermediate and can be explained by the Zimmerman Traxler principle.
Z-Enolate Produces Syn
Let us take a look at what happens in case of Z-enolate in order to produce the syn product more selectively.
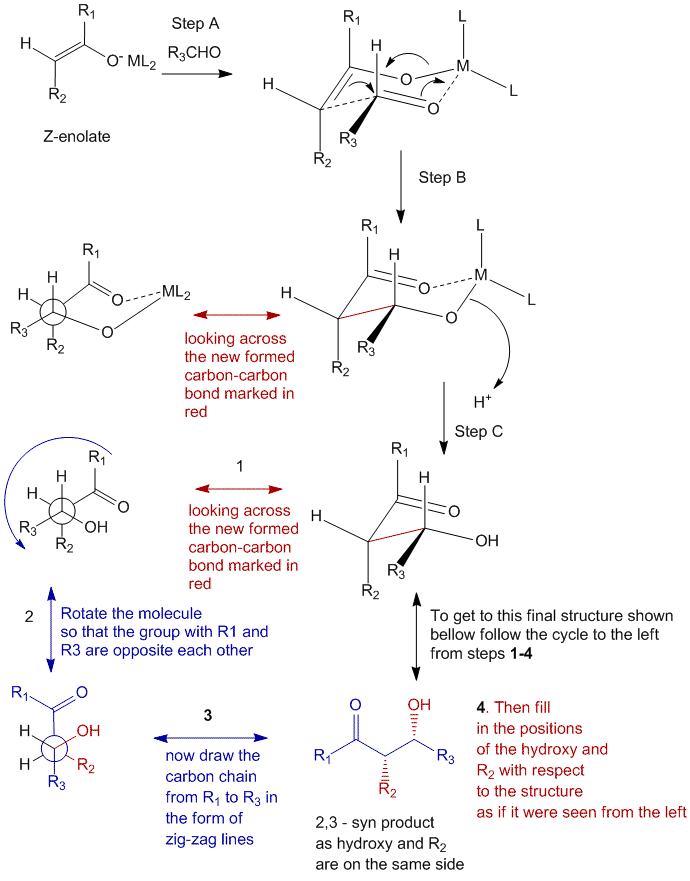
Step A
In step A, the Z-enolate is complexed with a metal (M) which has its ligands (L) attached to it and the aldehyde with the R3 group approaches the enolate complex to form a 6-membered cyclic transition state in a chair conformation. This is done in such a way so that if the R3 group is bulky, it will not be positioned axially, and thereby reduce the 1,3-diaxial interactions which can cause steric hinderances. It is due to this orientation and formation of the 6-membered cyclic transition state that the Z-enolate forms a selective product. Thus the bulkiness of the substituents R, is what matters when making the transition state.
Step B
In step B, the electrons shift over from the enolate to the aldehyde to form the new carbon-carbon bond. This bond thus formed gives rise to stereocenters.
Step C
In step C, the metal ligand complex is released to accept a proton which results in the formation of the β-alcohol.
In order to rewrite this chair conformation on a paper, it will be easier to represent it as a line-diagram (also known as the Natta projection) as shown as the equivalent in the figure below. However to achieve this, you must follow the side steps which are also shown in the diagram.
- Look across the new formed carbon-carbon bond and produce a Newman projection of the atoms involved.
- Rotate the molecule in Newman projection in such a way so as to keep the substituents with the R1 and the R3 groups opposite to each other (i.e. top and bottom in the above case).
- Draw the chain from R1 to R3 (by counting the atoms in between) in the form of zig zag lines, and add the carbonyl functional group of the enolate next to the R1.
- Fill in the positions of the hydroxy, and the R2 group with respect to the remaining structure as if it were seen from the left.
E-Enolate Produces Anti
In case of the E-enolate, the product formed is anti and this can be deduced in a similar chair conformation transition state as can be seen below.
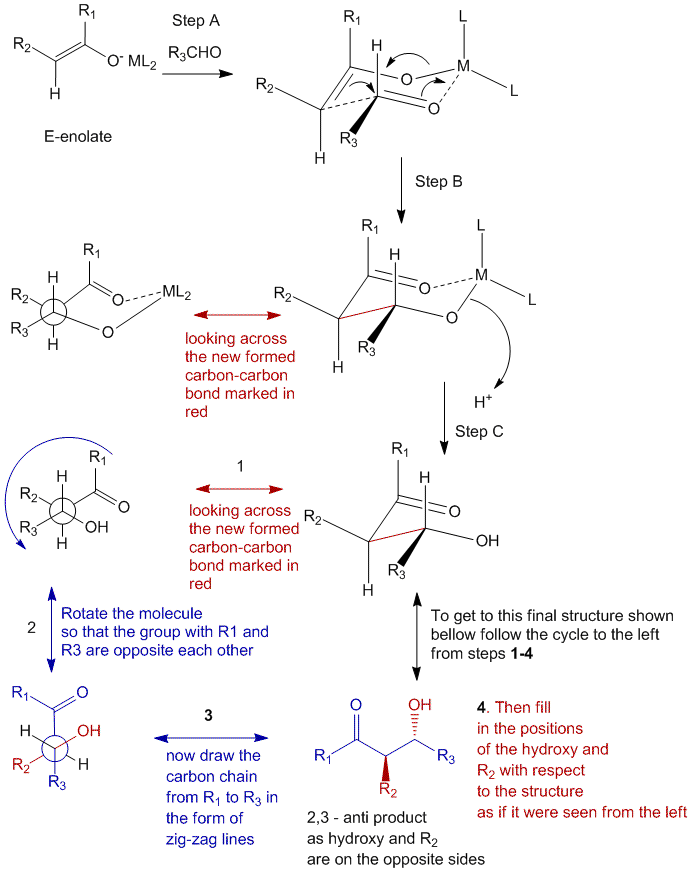
Step A
Similar to the Z-enolate, the E-enolate also forms a six-membered cyclic transition state with the metal complex and the aldehyde. In this case as well the R3 group of the aldehyde is placed equatorially in order to extinguish and 1,3-diaxial steric interactions. Once again, this cyclic intermediate is what controls the selective formation of the anti-product as will be seen ahead.
Step B
In step B, again there is a movement of electrons to form a carbon-carbon bond between the enolate and the aldehyde.
Step C
In step C, the metal ligand complex is lost and a proton is accepted to form the β-hydroxy carbonyl compound.
Once again in order to convert this structure into a more legible linear (Natta projection) structure you will have to go through the steps 1-4 which are shown to the left of the cycle which are:
- Look across the new formed carbon-carbon bond and produce a Newman projection of the atoms involved.
- Rotate the molecule in Newman projection in such a way so as to keep the substituents with the R1 and the R3 groups opposite to each other (i.e. top and bottom in the above case).
- Draw the chain from R1 to R3 (by counting the atoms in between) in the form of zig zag lines, and add the carbonyl functional group of the enolate next to the R1.
- Fill in the positions of the hydroxy, and the R2 group with respect to the remaining structure as if it were seen from the left.
Books on Organic Chemistry
Check out some of the best textbooks for Organic Chemistry
References
- Zimmerman, H. E.; Traxler, M. D. (1957). “The Stereochemistry of the Ivanov and Reformatsky Reactions. I”. Journal of the American Chemical Society, 79, 1920–1923.
- Advanced Organic Chemistry: Reactions and synthesis. By Francis A. Carey, Richard J. Sundberg