Aldol condensation reaction can be either acid catalyzed or base catalyzed. This page deals with the acid catalysis mechanism of the aldol reaction. Earlier, this reaction was thought to occur only with aldehydes. However, it has been realized that a similar reaction would occur with ketones and reactive carbonyl compounds with available α-hydrogens (the need for which will be apparent with the mechanism below).
The reaction proceeds with the condensation of an aldehyde (or carbonyl compound) with an enol. The product formed has an aldehyde (or carbonyl) group and a β-hydroxy (alcohol) group, giving the product the name aldol (or if the carbonyl compound is a ketone it maybe called a ketol). This condensation is often followed by spontaneous dehydration due to β-elimination to produce an α,β-unsaturated aldehyde or α,β-unsaturated ketone.
The mechanisms for acid catalyzed aldol condensation and base catalyzed aldol condensation is significantly different. While bases activate the nucleophile, acids activate the electrophile in the reaction.
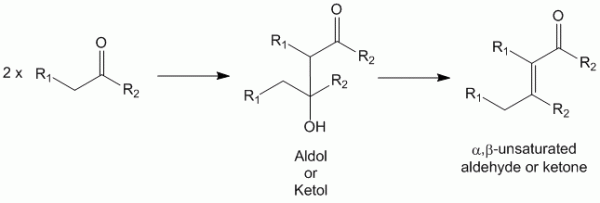
It must be noted that aldol condensation is an integral mechanism of Robinson annulation as well.
Mechanism of Acid Catalyzed Aldol Condensation
Step 1
In step 1 of the reaction, the acid acts as a proton donor and activates the carbonyl oxygen into a protonated form.
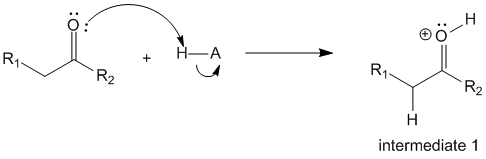
Step 2
In step 2, the intermediate 1 reacts with the conjugate base of the acid (i.e. A-) to produce the enol (intermediate 2).
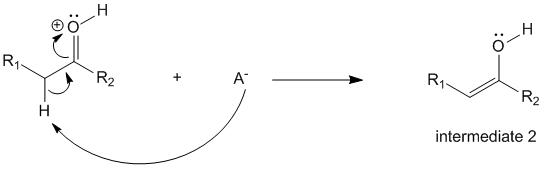
Step 3
This step involves the conjugation of the enol (intermediate 2) with another molecule of the activated carbonyl compound (intermediate 1) to produce the aldol (or ketol).
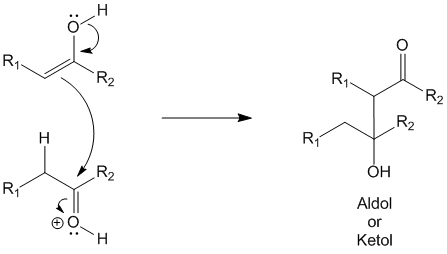
Step 4
In step 4, the aldol (or ketol) undergoes spontaneous dehydration due to base catalyzed dehydration to yield the α,β-unsaturated aldehyde or α,β-unsaturated ketone.
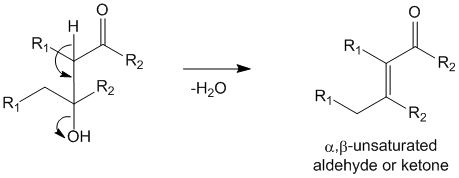
Books on Organic Chemistry
Check out some of the best textbooks for Organic Chemistry
References
- Advanced Organic Chemistry: Reactions and synthesis. By Francis A. Carey, Richard J. Sundberg
- Organic Chemistry, 6th Edition. By Robert T. Morrison and Robert N. Boyd
- Organic-Chemistry.org – http://www.organic-chemistry.org/namedreactions/aldol-condensation.shtm(accessed on June 29, 2011)