Click Here to Get Presentation
By: Preetpal S. Sidhu
Factor Xa is a zymogen of factor Xa, a serine protease, which occupies a pivotal position in the coagulation cascade. Factor Xa is found at the convergent point of extrinsic and intrinsic pathway leading to coagulation. Factor Xa and factor Va combine on the phospholipid membranes to form the “prothrombinase complex”, which activates prothrombin to thrombin. Thrombin cleaves fibrinogen to fibrin resulting in clot formation [1]. Factor Xa consist of two chains – a heavy chain and a light chain. These two are linked by a single disulfide bond. The light chain contains the N-terminal GLA (γ-carboxyglutamic acid) domain as well as two EGF (epidermal growth factor) domains. The GLA domain contains 11 γ-carboxyglutamic acid residues which are responsible for binding to the negatively charged phospholipids in the presence of calcium ions. The EGF domains provide the binding site for EPR-1 ( Effector cell protease receptor). The heavy chain contains trypsin-like serine protease domain. The organization of this domain is similar in coagulation enzymes like factor VIIa, factor Xa and protein C [2]. Factor Xa deficiency was first reported in 1950s in two families, Prower and Stuart; and as a result, factor Xa is also known as “Stuart” and “Stuart and Prower” factor [3].
CRYSTAL STRUCTURE: The first crystal structure of human factor Xa was deposited in May 1993. Till now total 191 crystal structures of factor Xa with various inhibitors have been deposited in the protein data bank. The active site of factor Xa is divided into four sub pockets as S1, S2, S3 and S4. The S1 subpocket determines the major component of selectivity and binding. The S2 sub-pocket is small, shallow and not well defined. It merges with the S4 subpocket. The S3 sub-pocket is located on the rim of the S1 pocket and is quite exposed to solvent. The S4 sub-pocket has 3 ligand binding domains, namely the “hydrophobic box”, the “cationic hole” and the water site. Factor Xa inhibitors generally bind in an L-shaped conformation, where one group of the ligand occupies the anionic S1 pocket lined by residues Asp189, Ser195, and Tyr228, and another group of the ligand occupies the aromatic S4 pocket lined by residues Tyr99, Phe174, and Trp228. Typically, a fairly rigid linker group bridges these two interaction sites [4].
Naturally occurring inhibitor of factor Xa was first reported in 1987. Tuszynski et al discovered antistasin, which was isolated from the extracts of Mexican leach, Haementeria officinalis [5]. Soon after this, another naturally occurring inhibitor, Tick anticoagulant peptide (TAP) was isolated from the extract of tick Ornithodoros moubata [6]. Synthetic factor Xa inhibitor can be of two types- one, which inhibits the generation of thrombin by blocking FXa directly (for example rivaroxaban, apixaban, otamixaban etc) and the other one, which indirectly inhibits FXa via a co-factor such as antithrombin [7].
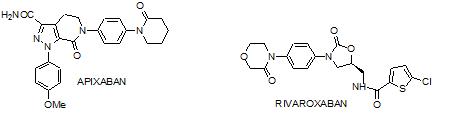
Rivaroxaban (BAY-59-7939) is an oral, direct, reversible, competitive, rapid, and dose-dependent inhibitor of FXa. Rivaroxaban inhibits FXa with more than 10,000-fold greater selectivity than other biologically relevant serine proteases, such as thrombin, trypsin, plasmin, factor VIIa, factor IXa, urokinase, and activated protein C. In addition to inhibition of free FXa in-vitro, rivaroxaban also inhibits FXa bound to the prothrombinase complex, raising the possibility that it could inhibit clot-bound FXa [7]. Only recombinant factor VIIa (rfVIIa) has been studied so far as a method of reversing anticoagulation induced by rivaroxaban [8]. The crystal structure of rivaroxaban shows that it forms two hydrogen bonds to Gly219 by the oxazolidinone core and NH group of the carboxamide, which provide the L-shape needed for FXa activity. In S1 pocket, key interaction involves chlorothiophene moiety, chlorine interacts with aromatic ring of Tyr228 located at the bottom of the S1 pocket. In the S4 pocket, nonpolar aryl ring extends across the face of Trp215 and morpholinone moiety is sandwiched between Tyr99 and Phe174 [9].
Apixaban is a follow-up compound of the oral, direct FXa inhibitor Razaxaban. Apixaban is a highly selective and potent (Ki=0.8nM) inhibitor of both free and prothrombinase bound FXa [10]. In X-ray crystal structure, p-methoxy group does not appear to interact with any specific residue in the S1 specificity pocket and is oriented in a planer manner. The pyrazole N-2 nitrogen atom interact with backbone of Gln192 and the carbonyl oxygen of carboxamide interact with NH of Gly216.The orientation of phenyllactum in the S4 region shows an edge to face interaction with Trp215 and is placed between the Tyr99 and Phe174 [11].
References:
- R. J. Leasdley. Coagulation Factor Xa Inhibition: Biological Background and Rationale, Current Topic in Med. Chem. 2001, 1, 151.
- K. Kamata, H. Kawomoto, T. Honma, T. Iwama, S. Kim, Structural Basis of Chemical Inhibition of Human Blood Coagulation Factor Xa, Proc. Natl. Acad. Sci., 1998, 95, 6630.
- T. P. Telfer, K. W. Denson, D. R. Wright, A New Coagulation Defect. Br. J. Haematol.,1956, 2, 308.
- R. Rai, P. A. Sprengeler, K. C. Elrod, W. B. Young, Perspectives on Factor Xa Inhibition. Current Med. Chem.2001, 8, 101.
- C. Dunwiddie, N. Thrnberry, H. Bull, M. Sardana, E. Simpson, Antistasin, a Leech-derived Inhibitor of Factor Xa. J. Biol. Chem., 1989, 264, 16694.
- L. Waxman, D. Smith, K. Arcuri, G. Vlasuk, Tick Anticoagulant Peptide is a Novel Inhibitor of Blood Coagulation Factor Xa. Science, 1990, 248, 593.
- M. P. Gulseth, J. Michaud, E. A. Nutescu, Rivaroxaban: An Oral Direct Inhibitor of Factor Xa, Am. J. Health-Syst Pharm, 2008, 65, 1520.
- E. Perzborn, J. Strassburger, A. Wilmen, In vitro and In vivo Studies of the Novel Antithrombotic Agent BAY 59-7939- an Oral, Direct Factor Xa Inhibitor, J. Thromb. Haemost. 2005, 3, 514.
- S. Roehrig, A. Straub, J. Pohlmann, T. Lampe, J. Pernerstorfer. Discovery of the Novel Antithrombotic Agent (BAY 59-7939): An Oral, Direct, Factor Xa Inhibitor, J. Med. Chem. 2005, 48, 5900
- J. Harenberg, M. Wehling, Current and Future Prospects for Anticoagulant Therepy: Inhibitors of Factor Xa and Factor IIa. 2008, 34, 39.
- D. Pinto, M. Orwat, S. Koch, K. Rossi, R. Alexander, A. Smallwood, P. Wong, A. Rendina, P. Lam, Discovery of 1-(4-Methoxyphenyl)-7-oxo-6-(4-(2-oxopiperidin-1-yl)phenyl)-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide(Apxiban, BMS-562247), a Highly Potent, Selective, Efficacious, and Orally Bioavailable Inhibitor of Blood Coagulation Factor Xa, 2007, 50, 5339.