Introduction
Cellular respiration involves 3 stages for the breakdown of glucose – glycolysis, Kreb’s cycle and the electron transport system. The total energy produced during glucose metabolism is described at Energetics of Cellular Respiration. We have seen the glycolysis pathway with animation previously. The Kreb’s cycle is named after Adolf Krebs who studied the utilization of oxygen in a pigeon. It is also commonly known as the citric acid cycle or the tricarboxylic acid cycle. Kreb’s cycle is a very important step in the metabolic pathway as it produces about 60-70% of ATP for release of energy in the body. It directly or indirectly connects with all the other individual pathways in the body too. It takes place in the mitochondria as all the enzymes and co-enzymes required are present there.
The Kreb’s Cycle occurs in two stages:
1. Conversion of Pyruvate to Acetyl CoA
Glycolysis of 1 molecule of glucose produces 2 molecules of pyruvate. Each pyruvate in the presence of pyruvate dehydrogenase (PDH) complex in the mitochondria gets converted to acetyl CoA which in turn enters the Kreb’s cycle. This reaction is called as oxidative decarboxylation as the carboxyl group is removed from the pyruvate molecule in the form of CO2 thus yielding 2-carbon acetyl group which along with the coenzyme A forms acetyl CoA.
The pyruvate dehydrogenase complex (PDH) comprises of three enzymes – pyruvate dehydrogenase, dihydrolipoyl transacetylase and dihydrolipoyl dehydrogenase each one playing an important role in the reaction as shown below. The PDH requires the sequential action of five co-factors or co-enzymes for the combined action of dehydrogenation and decarboxylation to take place. These five are TPP (thiamine phosphate), FAD (flavin adenine dinucleotide), NAD (nicotinamide adenine dinucleotide), coenzyme A (denoted as CoA-SH at times to depict role of -SH group) and lipoamide
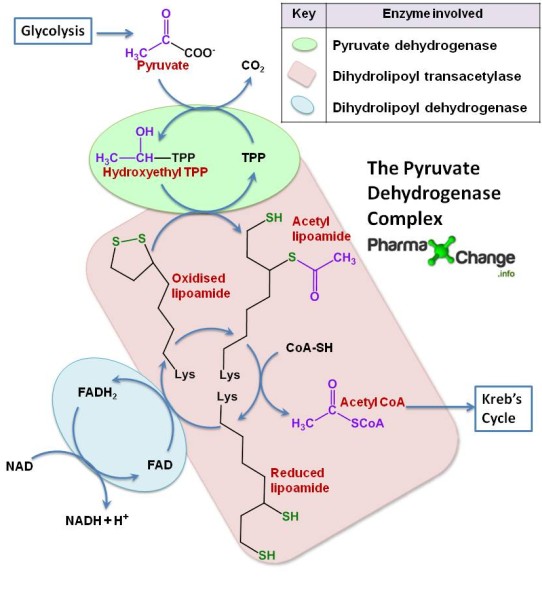
- Pyruvate reacts with the TPP (Thiamine Phosphate) bound part of pyruvate dehydrogenase and undergoes decarboxylation to give hydroxyethyl-TPP.
- This hydroxyethyl-TPP in turn gets oxidised to acetyl lipoamide by the same enzyme pyruvate dehydrogenase by the transfer of two electrons. These electrons then reduce the disulfide bond of the enzyme dihydrolipoyl transacetylase with the transfer of the acetyl group as highlighted in purple.
- Dihydrolipoyl transacetylase catalyses the transesterification forming acetyl CoA by transfer of acetyl group to coenzyme A.
- When acetyl CoA is being formed, at the same time reduced lipoamide is getting converted to oxidised lipoamide due to enzyme dihydrolipoyl dehydrogenase by the transfer of 2 hydrogen atoms to FAD.
- Dihydrolipoyl dehydrogenase transfers the reduced equivalents (2 hydrogen atoms) to FAD thus forming FADH2. FADH2 in turn transfers a hydride ion to NAD+ to form NADH+H+.
2. Acetyl CoA Enters the Kreb’s Cycle
The acetyl CoA produced from the pyruvate dehydrogenase complex enters the Kreb’s cycle.
The animation below describes the Kreb’s cycle in detail followed by the discussion. A static image of the cycle can be found next to the discussion for reference. Press the play button to progress in the animation.
Discussion
-
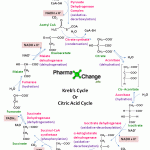
The Kreb’s Cycle or Citric Acid Cycle or Tricarboxylic Acid Cycle in a static image version of the animation. Acetyl CoA condenses with oxaloacetate (4C) to form a citrate (6C) by transferring its acetyl group in the presence of enzyme citrate synthase. The CoA liberated in this reaction is ready to participate in the oxidative decarboxylation of another molecule of pyruvate by PDH complex.
- Citrate is then isomerised to Isocitrate by the enzyme aconitase through the formation of the intermediate cis-aconitate. This is a reversible reaction as aconitase has an iron-sulfur center which can promote reversible addition of H2O to the double bond of enzyme-bound cis-aconitate in 2 different ways, one forming citrate and the other isocitrate.
- Isocitrate undergoes oxidative decarboxylation by the enzyme isocitrate dehydrogenase to form oxalosuccinate (intermediate- not shown) which in turn forms α-ketoglutarate (also known as oxoglutarate) which is a five carbon compound. CO2 and NADH are released in this step.
- α-ketoglutarate (5C) undergoes oxidative decarboxylation once again to form succinyl CoA (4C) catalysed by the enzyme α-ketoglutarate dehydrogenase complex. α-ketoglutarate dehydrogenase complex is similar to PDH complex and is made up of 3 enzymes and is dependent on five co-enzymes TPP, FAD, NAD, bound lipoate and conenzyme A. In this step once again NADH and CO2 are liberated. So in all 2 molecules of NADH and 2 molecules of CO2 is produced till now.
- Succinyl CoA is then converted to succinate by succinate thiokinase or succinyl coA synthetase in a reversible manner. This reaction involves an intermediate step in which the enzyme gets phosphorylated and then the phosphoryl group which has a high group transfer potential is transferred to GDP to form GTP. This GTP is converted to ATP by the enzyme nucleoside diphosphate kinase by donating its phosphoryl group to ADP. This reaction which involves the formation of GTP is a substrate level phosphorylation as it happens by using the energy formed by the oxidative decarboxylation of α-ketoglutarate.
- Succinate then gets oxidised reversibly to fumarate by succinate dehydrogenase. The enzyme contains iron-sulfur clusters and covalently bound FAD which when undergoes electron exchange in the mitochondria causes the production of FADH2.
- Fumarate is then by the enzyme fumarase converted to malate by hydration(addition of H2O) in a reversible manner.
- Malate is then reversibly converted to oxaloacetate by malate dehydrogenase which is NAD linked and thus produces NADH.
- The oxaloacetate produced is now ready to be utilized in the next cycle by the citrate synthase reaction and thus the equilibrium of the cycle shifts to the right.
Energetics of the Kreb’s Cycle
Keeping in mind that 1 molecule of glucose would produce 2 molecules of pyruvate via glycolysis. Hence the net energy produced by the Kreb’s cycle for each molecule of pyruvate is doubled for each molecule of glucose. Thus net energy yield in Kreb’s cycle can be summarized as follows for each molecule of glucose:
|
Reaction |
Number of ATP or reduced coenzyme formed* |
Number of ATP ultimately formed |
| 2 Pyruvate → 2 acetyl CoA |
2 NADH |
5 |
| 2 Isocitrate → 2 α- ketoglutarate |
2 NADH |
5 |
| 2 α- ketoglutarate → 2 succinyl CoA |
2 NADH |
5 |
| 2 Succinyl CoA → 2 succinate |
2 ATP |
2 |
| 2 Succinate → 2 fumarate |
2 FADH2 |
3 |
| 2 Malate → 2 oxaloacetate |
2 NADH |
5 |
|
TOTAL |
25 ATP |
* Note- This is calculated as 2.5 ATP per NADH and 1.5 ATP per FADH2. This is because there are multiple electron transport shuttle pathways through which these can be broken to ATP.
Regulation of Kreb’s Cycle
The amount of ADP and ATP largely control the citric acid cycle along with the activity of three key enzymes within the cycle:
- Availability of ADP: ADP is a key substrate which finally gets converted to ATP that is essential for the energetics of the cell. A drop in ADP levels would result in inhibition of the electron transport system leading to accumulation of NADH and FADH2. These in turn inhibit the enzymes below.
- Citrate Synthase: inhibited by ATP, acetyl CoA, NADH, and succinyl CoA.
- Isocitrate Dehydrogenase: activated by ADP, and inhibited by NADH and ATP.
- α-ketoglutarate dehydrogenase: inhibited by NADH and succinyl CoA.
Recommended Texts
- David L. Nelson and Michael M. Cox, Lehninger Principles of Biochemistry 6th Edition
- Jeremy M. Berg, John L. Tymockzo and Luber Stryer, Biochemistry 7th Edition
- Reginald H. Garrett, Charles M. Grisham, Biochemistry by Reginald H Garrett 5th Edition
.
- U. Satyanarayana, U. Chakrapani, Biochemistry by U. Satyanarayana. 3rd Edition.

I am b.pharm student. Secondary super for biochemistry notes
Amazing presentation of biochemical pathways. From the point of view of a current medical student the step by step animations have been wonderful. Thank you for your work!
Excellent and simplified explanation especially on how ATP is generated. How does the TCA cycle contribute to HCO3 synthesis?