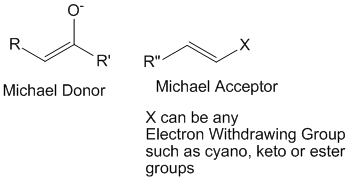
Michael addition is a well known name reaction which involves a michael donor – generally an enolate (a nucleophile), and a michael acceptor – generally a conjugated system with an electron withdrawing group such as cyano, keto or ester (an electrophile).
The reaction generally proceeds under base catalysis as will be shown in the following mechanism. Michael addition is a great tool which can be used by organic chemists to increase the number of carbons of a molecule and make larger molecules by formation of new carbon-carbon bonds.
Mechanism
Step 1
In the first step, an enolate (or michael donor) is produced in the precence of a base by the abstraction of the alpha hydrogen of the carbonyl containing compound.
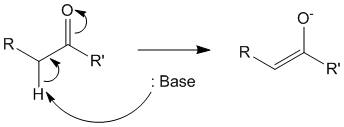
Step 2
In step 2, the michael donor (enolate) reacts with the michael acceptor to produce the molecule as shown. This leads to formation of a new carbon-carbon bond.
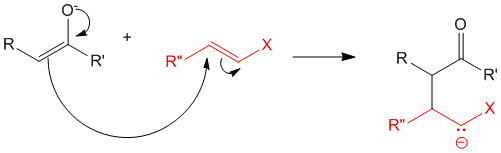
Step 3
In the final step, there is regeneration of the base catalyst by the carbanion formed in step 2 to form the final product.

Example of Michael Addition
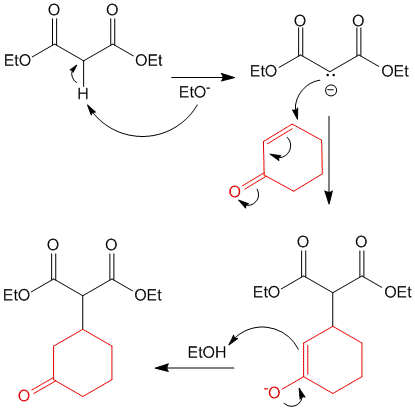
A very good example of Michael Addition is the reaction of diethyl malonate (Michael Donor) with cyclohexenone (Michael Acceptor) in the presence of base sodium ethoxide. The reaction proceeds as shown and joins the two molecules by formation of a carbon-carbon bond.
Books on Organic Chemistry
Check out some of the best textbooks for Organic Chemistry
References
- Advanced Organic Chemistry: Reactions and synthesis. By Francis A. Carey, Richard J. Sundberg
- Organic Chemistry, 6th Edition. By Robert T. Morrison and Robert N. Boyd
- Organic-Chemistry.org – http://www.organic-chemistry.org/namedreactions/michael-addition.shtm (accessed on April 24, 2011)