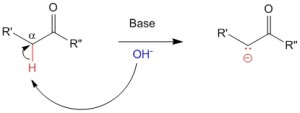
Phenolates are formed when phenolic hydroxy groups are exposed to basic conditions.
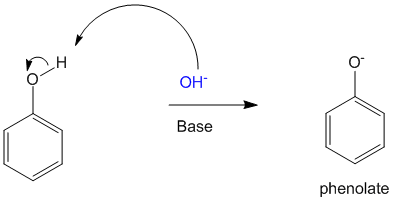
Generally even weak bases are capable of forming the phenolate anion.
This phenolate anion can be reacted with an alkyl halide to yield a possible two products- either the O-alkylated (product 1) or the C-alkylated (product 2).
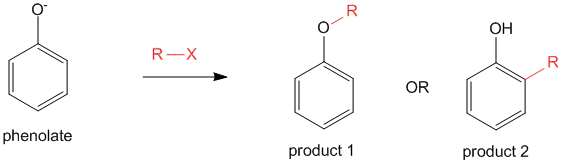
The product 1 is formed by the O-alkylation of the phenolate anion, while product 2 is formed via C-alkylation of the phenolate anion. The possibility of product 1 being formed is higher than the probability of formation of product 2. However, under certain conditions, it is possible to form product 2 as well.
NOTE – The O-alkylated product is similar to the reaction seen in Williamson Ether Synthesis
Mechanism
Step 1
In the presence of base the phenol gets converted to the phenolate anion

Step 2
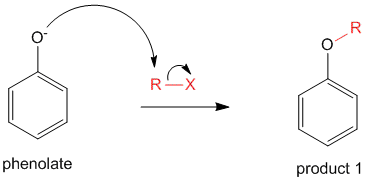
This phenolate anion can undergo O-alkylation via the normal SN2 reaction to yield the product 1.
Alternatively, the phenolate anion can undergo a resonance transformation and then undergo a C-alkylation with the alkyl halide. The phenol is reformed to restore aromaticity of the phenyl ring yielding product 2.
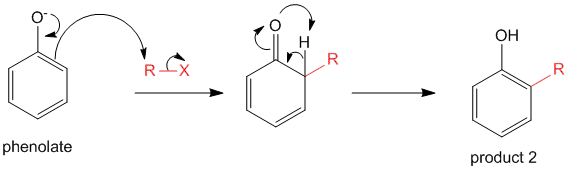
As aromaticity is disrupted in this mechanism, the energy barrier for the intermediate is higher, and therefore, the probability of product 2 is lower. However if conditions are tweaked such that the oxygen anion is blocked, the possibility of product 2 increases.
Conditions for O-alkylation vs C-alkylation of Phenolates
The solvent selection widely controls what products you get for this reaction.
- For O-alkylation: Use DMF, DMSO, t-Butanol
- For C-alkylation: Use of protic solvents like water or Trifluoro Ethanol (TFE)
Use of protic solvents pushes the formation of C-alkylated product as the solvent is capable of hydrogen bonding with the phenolate oxygen and therefore shields the oxygen, thus preventing it from attacking the alkyl halide.
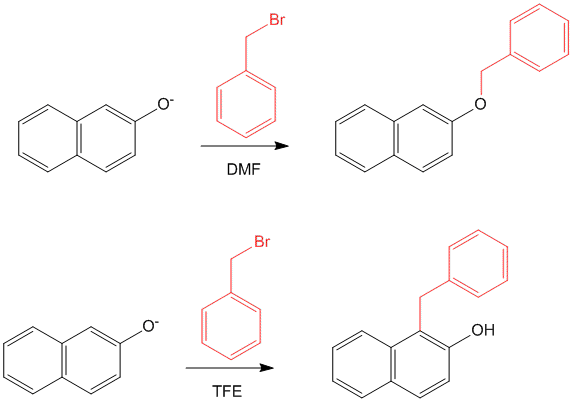
For example,
2-Naphthol or β-Naphthol, reacts with benzyl bromide in the presence of base and DMF to give O-alkylated product, while in the presence of trifluoro ethanol (TFE) it yields the C-alkylated product.
Books on Organic Chemistry
Check out some of the best textbooks for Organic Chemistry
References
- Advanced Organic Chemistry: Reactions and synthesis. By Francis A. Carey, Richard J. Sundberg