Introduction:
Proteins are an integral part of the human body and are involved in many of the biosynthetic pathways. An average of 300-400 grams of protein is being digested, degraded and being synthesized every day in the human body. Out of all the amino acids (20) present glutamate and glutamine play an essential role and contribute to around 50% of the amino acid pool. Amino acid pool is nothing but the free amino acid present in the circulation system at a certain point of time that adjusts to meet the body’s need for amino acid and proteins. The input i.e sources that contribute amino acids (dietary protein:40-100 grams/day and cellular protein as well as the non-essential amino acids produced by the body) and the output i.e utilization of amino acids (to produce energy or to synthesize important nitrogenous compounds) help in maintaining the amino acid pool. Before the protein reaches the liver for metabolism, they are digested from food and broken down to their respective amino acids in the gastrointestinal tract. Amino acid metabolism then takes place in the liver once transferred via blood from the intestine.
Digestion of dietary proteins in gastrointestinal tract (GI tract)
In humans, proteins get degraded to their respective amino acids in the gastrointestinal tract.
Note- All zymogens in the pathway below have a “-gen” suffix in the name. Hormones are highlighted in blue, organs in red and enzymes in green.
- Hormone gastrin is released due to the stimulation of gastric mucosa by the entry of protein from the diet into the stomach which in turn stimulates the secretion of pepsinogen (zymogen) by the chief cells of gastric gland and HCl by the parietal cells. The acidic environment produced now has two roles to play,it acts bactericidal and thereby killing most bacteria and other foreign cells and it also denatures the globular proteins by unfolding them and making the peptide bonds more susceptible to enzymatic hydrolysis.
- The released inactive pepsinogen (zymogen) is then converted to its active form pepsin by pepsin itself which hydrolyses the the peptide bonds of the proteins leaving behind smaller fragments of smaller peptides.
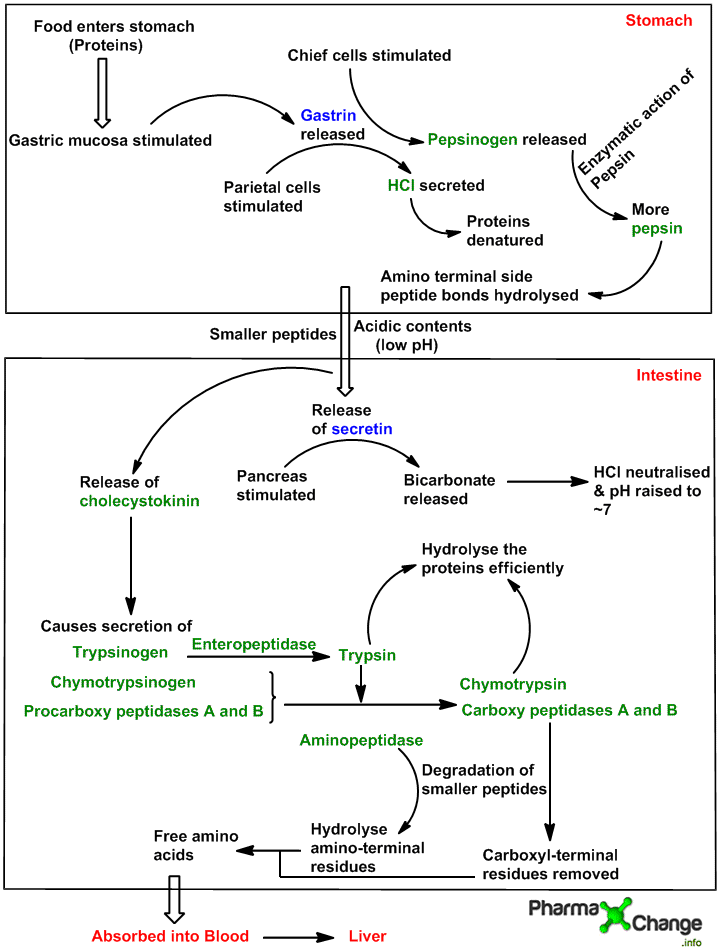
- These smaller peptides now enter the small intestine where they are further digested by the release of hormone secretin (activated due to the low pH) in the blood.
- Here the pancreas are stimulated by the hormone secretin released. On stimulation pancreas release bicarbonate to neutralize the acidity produced by HCl and thereby raising the pH to around 7. This neutralization is important for the other enzymes which would otherwise be denatured due to the acidic pH of the stomach.
- The contents are then further are passed to upper intestine where the hormone cholecystokinin is activated and in turn stimulates the release of other zymogens (trypsinogen, chymotrypsinogen, procarboxypeptidase A and B).
- Enteropeptidases present in the small intestine convert trypsinogen to trypsin, which in turn activates further conversion of trypsinogen to trypsin in the intestine.
- Chymotrypsinogen, procarboxypeptidase A and B are also activated by trypsin.The active forms trypsin and chymotrypsin produced through a chain of reactions from trypsinogen and chymotrypsinogen, further hydrolyse the smaller peptides.
- As all the enzymes (trypsin, chymotrypsin and pepsin) have different specificities for different amino acids, digestion is done very systematically and efficiently.
Note- The pancreas protects itself from digestion by the proteolytic enzymes by releasing the pancreatic trypsin inhibitor. - Carboxypeptidase A and B catalyze the removal of the carboxyl group whereas hydrolysis of the amino end is carried out by aminopeptidase thus further degrading these smaller peptides. Then the respective free amino acids formed by digestion enter the blood capillaries from the epithelial cell lining and are transported to the liver where they are metabolized.
Recommended Texts
- David L. Nelson and Michael M. Cox, Lehninger Principles of Biochemistry 6th Edition
- Jeremy M. Berg, John L. Tymockzo and Luber Stryer, Biochemistry 7th Edition
- Reginald H. Garrett, Charles M. Grisham, Biochemistry by Reginald H Garrett 5th Edition
.
- U. Satyanarayana, U. Chakrapani, Biochemistry by U. Satyanarayana. 3rd Edition.


Is there any way at all that cow’s milk protein in a mother’s diet could be found in her breast milk causing allergy in her baby?
Highly doubtful, our digestive systems are equipped to disintegrate proteins to amino acids which get absorbed. So milk protein would get hydrolyzed to amino acids which get absorbed, and it is highly unlikely that anyone is allergic to amino acids which are essential for healthy living.
its very informative
Hello there, simply was aware of your weblog thru Google, and located that it’s truly informative. I will be grateful in case you continue this in future. A lot of other folks will likely be benefited out of your writing. Cheers!
that was a beautiful explanation. I thought of that too
I thought hcl helps in conversion of pepsinogen to pepsin
Hi Peter,
That comment is partially correct. HCl does help in activation of pepsinogen to pepsin but not in a direct manner. Hydrochloric acid just creates an acidic environment allowing the pepsinogen to unfold and cleave itself (autolysis) to get activated to pepsin. After this the pepsin itself activates more pepsinogen to enhance the amount of pepsin present. So all HCl does is creates the environment for autolysis of pepsinogen, while the major chunk of pepsin produced is due to pepsin itself. Reference: Katherine S et. al. Crystal Structure of Human Pepsinogen A. Advances in Experimental Medicine and Biology Volume 436, 1998, pp 259-263.