Introduction
Thin layer chromatography (TLC) is a technique which is extensively used by organic chemists to monitor reactions and can also be useful in identification of products, as well as functional groups. This article aims to take a look at the principle of thin layer chromatography and to better explain it using an animation.
Principle of Thin Layer Chromatography (TLC)
In the thin layer chromatography (TLC) analysis, a glass plate is coated by adsorbent materials such as silica. Silica is the most commonly used adsorbent material for TLC analysis. Structurally silica gel consists of a matrix of Si-OH groups which can interact with molecules via hydrogen bonding and adsorption. A few microliters of a dilute solution is put onto the silica surface of the plate using a microcapillary. This is known as “spotting”. The plate is then placed in a jar containing a solvent (generally mixtures of ethylacetate and hexanes). As time goes, the solvent gradually rises up the plate due to capillary action carrying the components of the sample with it.
Different molecules are carried up the plate to different distances due to variable interactions with the adsorbent material. For example, when silica is used, polar molecules with groups such as hydroxy (OH) or amine (NH2) will tend to form hydrogen bonds with the silica matrix (Si-OH) groups and will therefore not move as fast up the plate. While relatively non-polar molecules will have fewer interactions with the matrix and will tend to be more soluble in the solvent phase and therefore rise faster up with the solvent front (see animation below)
Once the solvent has risen a particular distance sufficient to separate components of the spot, the plate is either visualized directly using ultraviolet light, or it is developed using a stain to check for specific types of molecules.
Animation of Thin Layer Chromatography (TLC)
The animation below will demonstrate what exactly happens in a thin layer chromatography analysis. Press the play button in order to move ahead in the animation. To rewind the animation move the scroll bar to the left
[swfobject]2340[/swfobject]
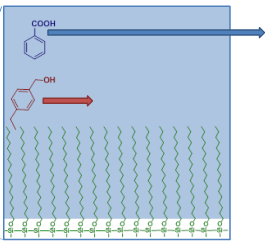
In the animation, a spot containing a mixture of toluene (-CH3 containing non polar molecule) and phenol (-OH containing polar molecule) is spotted on a TLC plate coated with silica (Si-OH) particles.
The plate is then placed in a developing jar (or beaker) containing a particular solvent system (something like 50:50 ethylacetate:hexanes).
The solvent gradually rises up the plate taking with it the two components to different levels. The polar molecule phenol is retained on the surface due to possible hydrogen bonding interactions with the silica, and hence does not rise with the solvent front as quickly. While the non-polar molecule toluene is quick to rise up with the solvent front as it makes little or no interaction with the silica matrix.


Thanks for explaining principle of tlc in very simple language
very good material easy to understood
Its realy useful and easy to understand, animaion is well suited wih topic.
Many thanks
Hemant
thanks for the useful article. im getting ready for my analycal chem exam and thats exactly what i was looking for 🙂
its very good website for all types tpoics with all students very helpful .
Thanks so much ,,,really helped in helping me understand TLC……………………………….
Tanks so much, really helped in helping me understand TLC,