The Henderson-Hasselbalch equation is extremely useful in the fields of pharmacy, pharmaceutical sciences, chemistry, biology and biochemistry. The derivation of the Henderson-Hasselbalch equation is therefore significant for any student of the sciences.
Derivation of Henderson-Hasselbalch Equation
For Acids
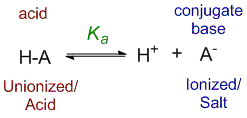
According to the law of dissociation, the acid dissociation constant Ka can be defined by the equation:
Ka = [H+][A–]/[HA]
where [H+] and [A–] are the concentrations of the ionized form of the acid, while [HA] is the concentration of the unionized form.
Taking log on both sides you get:
log Ka = log ([H+][A–]/[HA])
Splitting the log terms into separate components gives you:
log Ka = log [H+] + log ([A–]/[HA])
Since pKa = -log10 Ka and pH = -log10 [H+], therefore:
-pKa = – pH + log ([A–]/[HA]
Rearranging the terms we get the Henderson-Hasselbalch equation for acids as:
pH = pKa + log ([A–]/[HA])
This equation can also be written as:
pH = pKa + log [(salt)/(acid)]
or
pH = pKa + log ([ionized]/[unionized])
Similarly for bases:
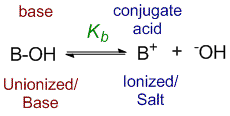
According to the law of dissociation, the base dissociation constant Kb can be defined by the equation:
Kb = [OH–][B+] / [BOH]
where [B+] and [OH–] are the concentrations of the ionized form of the base, while [BOH] is the concentration of the unionized form.
Taking log on both sides you get:
log Kb = log ([OH–][B+] / [BOH])
Splitting the log terms into separate components gives you:
log Kb = log [OH–] + log ([B+] / [BOH])
Since pKb = -log10 Kb and pOH = -log10 [OH–], therefore:
-pKb = – pOH + log ([B+]/[BOH])
Rearranging the terms we get the Henderson-Hasselbalch equation for bases:
pOH = pKb + log ([B+]/[BOH])
This equation can also be written as:
pOH = pKb + log [(salt)/(base)]
or
pOH = pKb + log ([ionized]/[unionized])
pKa And pH Can Be Used Universally For Both Acids and Bases
Generally the term pKa can be used to measure the strength of an acid and pKb for bases. However, in order to compare acidity with basicity the pKa of a base may also be calculated in order to keep a single scale of comparison. To derive the relationship for bases in terms of pH and pKa we must look at the relationship between pKa and pKb as well as the relationship between pH and pOH.
The product of Ka and Kb is the dissociation constant of water:
Ka . Kb = Kw = [H+][OH–]
Since [H+][OH–] = Kw = 10–14, therefore:
Ka . Kb = 10–14
Therefore taking logs:
log Ka + log Kb = log 10–14
thus, since pKa = -log Ka and pKb = -log Kb:
pKa + pKb = 14
and it is known that
[H+] [OH–] = Kw = 10-14
taking logs:
log [H+] + log [OH–] = log 10-14
Since pH = -log [H+] and pOH = -log [OH–]
pH + pOH = 14
Thus for bases, the Henderson-Hasselbalch equation:
pOH = pKb + log ([B+]/[BOH]) may be rewritten as:
14 – pH = 14 – pKa + log ([B+]/[BOH])
subtracting 14 on both sides and multiplying both sides by -1 we get the Henderson-Hasselbalch equation for bases in terms of pKa and pH:
pH = pKa – log ([B+]/[BOH])
or
pH = pKa – log ([salt]/[base])
or
pH = pKa – log ([ionized]/[unionized])
The pKa Scale for Acidity and Basicity

It must always be kept in mind that acidity and basicity are relative terms. These terms were made to judge whether a reaction would go forward or not. The pKa value of acids and bases gives an idea of how strong the acids or bases are. The pKa of water is 15.7. Any value higher than this pKa value increases basicity, while values which decrease from this value are increasing in acidity. For example, the pKa values for strong acids such as HCl can even go into the negative values with a pKa of -8. While, a strong base such as ammonia (NH3) has a pKa of 38.
What Information the Henderson-Hasselbalch Equation Gives You?
Check out the applications and examples of using Henderson-Hasselbalch equation. The equation can basically tell you the following:
1] How acidic or basic the molecule is? i.e. What is the pKa of the molecule?
The Henderson-Hasselbalch equation lets you calculate the pKa of a molecule using simple experimental protocols. As we have seen above, the pKa gives an idea of the acidity and basicity of molecules, and is hence very useful in pharmaceutical, chemistry, biology and biochemistry fields.
2] If a molecule is placed in a solution of particular pH, what fraction of the molecule will be ionized vs unionized?
The Henderson-Hasselbalch equation gives you information of the abundance of ionized and unionized fractions of molecules within a given solution. This is useful for certain reactions as well as important for absorption, distribution and excretion of drugs. Hence it is very useful in fields of chemistry, biochemistry, and pharmacy.
3] Predict the pH of the solution?
The Henderson-Hasselbalch equation is useful in estimating the pH of buffer solutions as can be seen in the applications and examples page.
Check Out:
- pKa table and its use
- Applications and example roblems based on Henderson-Hasselbalch equation

first ionized salt is used but in the end it is written ionized/unionized . why is that ?
can the equation be used to measure the metabolic acidosis and alkalosis?
Yes it can.
So if I have a weak avid with a pKa 7.4 and pH assumed to be 6.4
I would have a 10:1 ratio
So 90 percent of acid is absorbed and 10 percent excreted???
But what if I it’s reversed and I get 1:10 ratio
Does that mean 90 acid is excreted and 10 absorbed????
Hi LjK0001,
The absorption and excretion will depend on the environment in which the molecule is placed. Check out the sample problems of Henderson Hasselbalch equation to get a better idea