Alkaloids are basic nitrogen containing compounds. They are generally obtained from plants, animals and microorganisms and often demonstrate a marked physiological action. Alkaloids show greatly diverse structure and origins as well as pharmacological action. The only thing that unites all these natural compounds under the term ‘alkaloids’ (alkali-like) is the nitrogen atom which is present in all of them. According to pharmacognosy, alkaloids are broadly classified into two classes depending upon whether the nitrogen is a part of a ring or not.
1] Non-Heterocyclic Alkaloids or Atypical Alkaloids:
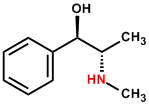
These are also sometimes called proto-alkaloids or biological amines. These are less commonly found in nature. These molecules have a nitrogen atom which is not a part of any ring system. Examples of these include ephedrine, colchicine, erythromycin and taxol etc. Table below shows the chemical structure and biological significance of these compounds:
2] Heterocyclic Alkaloids or Typical Alkaloids:
Structurally these have the nitrogen as a part of a cyclic ring system. These are more commonly found in nature. Heterocyclic alkaloids are further subdivided into 14 groups based on the ring structure containing the nitrogen.
| No. | Heterocycle | Example |
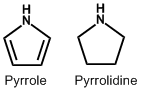
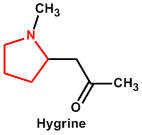
| 1. | Pyrrole and Pyrrolidine | Hygrine, Stachydrine |
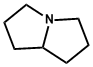
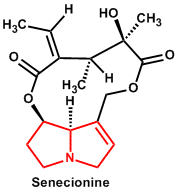
| 2. | Pyrrolizidine | Senecionine, Symphitine, Echimidine, Seneciphylline |
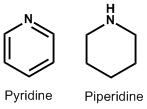
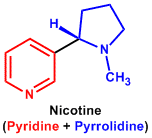
| 3. | Pyridine and Piperidine | Lobeline, Nicotine, Piperine, Conine, Trigonelline |
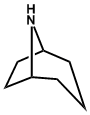
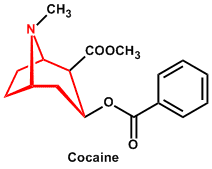
| 4. | Tropane (piperidine/N-methyl-pyrrolidine) | Cocaine, Atropine, Hyoscyamine, Hyoscine |
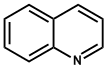
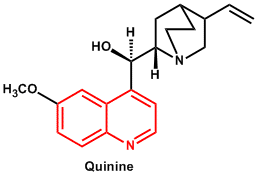
| 5. | Quinoline | Quinine, Quinidine, Cinchonine, Cinchonidine |
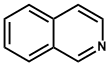
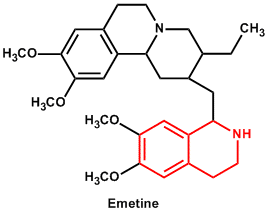
| 6. | Isoquinoline | Morphine, Emetine, Papaverine, Narcotine, Tubocurarine, Codeine |
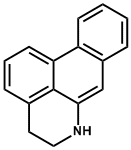
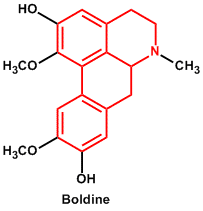
| 7. | Aporphine (reduced isoquinoline/naphthalene) | Boldine |
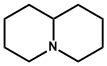
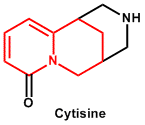
| 8. | Quinolizidine | Lupanine, Cytisine, Laburnine, Sparteine |
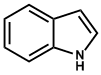
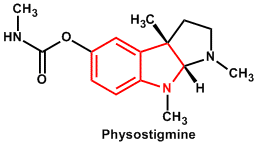
| 9. | Indole or Benzopyrole | Ergometrine, Vinblastine, Vincristine, Strychnine, Brucine, Ergotamine, Yohimbine, Reserpine, Serpentine, Physostigmine |
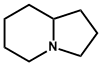
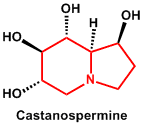
| 10. | Indolizidine | Castanospermine, Swainsonine |
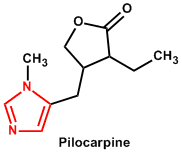
| 11. | Imidazole or glyoxaline | Pilocarpine, Pilosine |
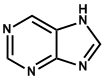
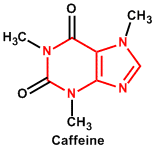
| 12. | Purine (pyrimidine/imidazole) | Caffeine, Theobromine |
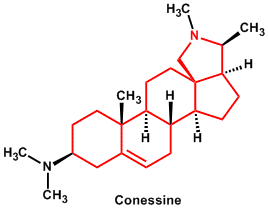
| 13. | Steroidal (some combined as glycosides)* | Conessine, Solanidine |
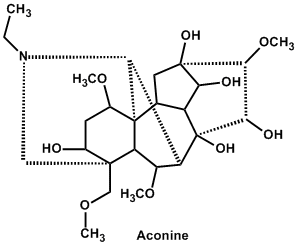
| 14. | Terpenoid* | Aconitine, lycaconitine, Aconine |
*Note- Steroidal and terpenoid classes are also treated as separate classes or along with glycosides.
Other methods of classification of alkaloids include:
- Based on their pharmacological action: Alkaloids have very diverse pharmacological actions. They are known to be adrenergics, antibiotics, poisons, stimulants, diuretics, astringents, anti-inflammatory, anti-hypertensives, anti-mydriatics, analgesics, anti-gout, expectorant, emetic, anti-spasmodic and many others. However, structurally diverse molecules may show similar pharmacological actions while in certain cases the activity might be identical for specific structure. Hence it is very difficult to classify them on the sole basis of pharmacological action.
- Based on their taxonomy: Alkaloids can be classified on the basis of the biological source from which they are obtained but this generalizations do not work most often.
- Based on their biosynthetic origin: Biosynthetic origin here means from which fundamental chemical building block these alkaloids are derived. For example indole alkaloids often come from tryptophan, pyrrolidine and tropane containing alkaloids come from proline and ornithine, quinolizidine containing alkaloids are derived from lysine. Biosynthetic origin of classification is beneficial in a more ordered classification, however, it is often unknown how most of the alkaloids are synthesized by the plants.
References
- Evans, W. C. Trease and Evans Pharmacognosy, 16th ed.; Elsevier: New York, 2009.
- Kokate, C. K.; Gokhale, S. B.; Purohit, A. P. A textbook of Pharmacognosy, 29th ed.; Nirali Prakashan: Pune, 2009.

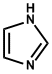

Excellent sir
Excellent
this was so nice……………………i liked it thanks……………………………