CLICK HERE TO DOWNLOAD THE PRESENTATION – FLUORESCENT LABELING OF BIOMOLECULES WITH ORGANIC PROBES
Abstract:
Biomolecules are organic compounds that are involved in the maintenance and metabolic processes of living organisms. Amino acids, peptides and proteins are extremely important biomolecules that play a vital role in the human body. Many disease conditions are due to impaired amino acid metabolism (e.g. phenylketonuria). An important class of therapeutic agents is the peptide based pharmaceuticals (e.g. interferon) that have been used to treat the disease multiple sclerosis. These biomolecules can be detected by tagging them with a fluorescent label. The resulting conjugate or complex will show fluorescence, thereby facilitating their detection.1-3
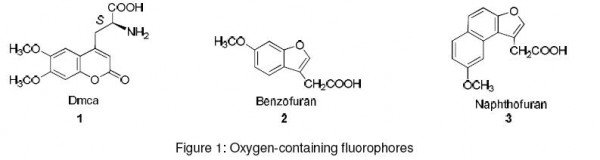
Coumarins are an important class of oxygen-containing fluorophores commonly used for labeling amino acids. Dmca (1) is a coumarin-containing un-natural amino acid. It has an absorption maximum of 345 nm, emission maximum of 440 nm and ΦF (fluorescent quantum yield) of 0.52.3 Furans (Figure 1) such as benzofuran (2) and naphthofuran (3) are another important class of oxygen- containing fluorophores. These can be linked to L-amino acids to form fluorescent derivatives. Naphthofuran has ΦF of 0.20 and shows a bathochromic shift of 13 nm in the absorption spectra and 34 nm in the emission spectra as compared to benzofuran, which has ΦF of 0.020. On this basis it was concluded that naphthofuran is a better fluorescent label than benzofuran.4,5
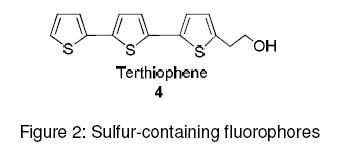
Oligothiophenes like terthiophene (Figure 2) are sulfur-containing compounds that have intrinsic fluorescence. By phosphoramidite coupling, they are used to tag oligonucleotides.6 Oligothiophene functionalized with N-succinimidyl esters have been conjugated to oligonucleotides at the 3’ end and a fluorescence quencher, dabcyl, at the 5’ end. Fluorescence Resonance Energy Transfer (FRET) experiments are then carried out to monitor the hybridization of the resulting fluorescent oligonucleotides to complementary single-stranded DNA target.7,8
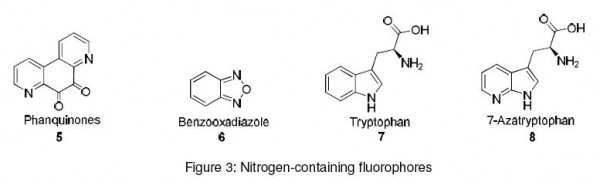
Most amino acids are not readily detectable due to the absence of a strong chromophore orfluorophore. Fluorogenic derivatization prior to HPLC analysis of amino acids serves as an efficient tool in the analysis of amino acids. Phanquinones (5) and benzooxadiazoles (6) are nitrogen containing fluorophores that can be used as pre-column derivatization agents. These compounds aredevoid of intrinsic fluorescence. However, on conjugation with amino acids, they form the corresponding fluorescent conjugates.9-11
Tryptophan (7), an amino acid with intrinsic fluorescence, has been widely studied. When this amino acid was used as a fluorescent probe, a number of problems occurred. Some of them were fluorescence quenching due to neighboring protonated acidic groups and also difficulty in detection if more than one tryptophan residue is present. Such problems were overcome by using the non-coded analog of tryptophan. The non-coded analog, 7-azatryptophan (8) is used to investigate the disulfide coupled folding of the hirudin 1-47 fragment by FRET experiments.12
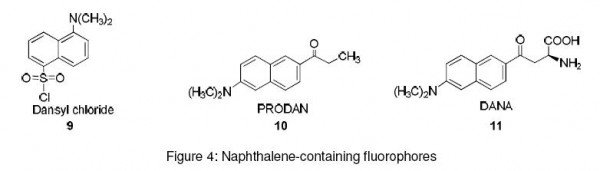
Naphthalene ring-containing fluorophores have been extensively used as pre-column derivatization agents for the analysis of amino acids in biological samples. Dansyl chloride (9) has been used in the “in-synthesis” labeling of peptides. Here, solid phase method is used for peptide synthesis. Dansylchloride is added to the growing peptide chain, thus resulting in a fluorescent peptide.13 6-Propionyl-2-(dimethylamino)naphthalene (PRODAN) (10) and 6-(2-dimethylaminonaphthoyl)alanine (DANA) (11) are environment-sensitive fluorophores. Their spectral characteristics change with the environment.14,15 DANA utilizes its unique sensitivity to the environment in monitoring the phosphorylation-dependent binding of peptides to proteins.16
In summary, biomolecules are mediators of various physiological processes as well as an important class of therapeutic agents and thus need to be detected and analyzed. Fluorescent labeling is an efficient method that can be used for bioanalytical purposes. These organic fluorophores have been widely used for labeling and subsequent detection of biomolecules.
References:
1. Goncalves, M. S. T. Fluorescent labeling of biomolecules with organic probes. Chem. Rev. 2009, 109,190-212.
2. Bennett, F. A.; Barlow, D. J.; Dodoo, A. N. O.; Hider, R. C.; Lansley, A. B.; Lawrence, M. J.; Marriott, C.;Bansal, S. S. Synthesis and properties of (6,7-dimethoxy-4-coumaryl)alanine: a fluorescent peptide label. Anal. Biochem. 1999, 270, 15-23.
3. Wang, W.; Li, H. An efficient synthesis of the intrinsic fluorescent peptide labels, (S)- and (R)-(6,7-dimethoxy-4-coumaryl)alanines via asymmetric hydrogenations. Tetrahedron Lett. 2004, 45, 8479-8481.
4. Piloto, A. M.; Costa, S. P. G.; Goncalves, M. S. T. A naphthol[2,1-b]furan as a new fluorescent label:synthesis and spectral characterization. Tetrahedron Lett. 2005, 46, 4757-4760.
5. Piloto, A. M.; Fonseca, A. S. C.; Costa, S. P. G.; Goncalves, M. S. T. Carboxylic fused furans for aminoacid fluorescent labeling. Tetrahedron Lett. 2006, 62, 9258-9267.
6. Capobianco, M. L.; Naldi, M.; Zambianchi, M.; Barbarella, G. Oligothiophene phosphoramidites for oligonucleotides labeling. Tetrahedron Lett. 2005, 46, 8181-8184.
7. Barbarella, G.; Zambianchi, M.; Sotgiu, G.; Ventola, A.; Galeotti, M.; Gigli, G.; Cazzato, A.; Capobianco,M. L. Thiophene-based fluorescent markers for the efficient labeling of monoclonal antibodies and oligonucleotides. J. Non-Cryst. Solids. 2006, 352, 2465-2467.
8. Barbarella, G.; Zambianchi, M.; Ventola, A.; Fabiano, E.; Sala, F.; Gigli, G.; Anni, M.; Bolognesi, A.;Polito, L.; Naldi, M.; Capobianco, M. Bright oligothiophene N-succinimidyl esters for efficient fluorescent labeling of proteins and oligonucleotides. Bioconjugate Chem. 2006, 17, 58-67.
9. Gatti, R.; Gioia, M. G.; Di Pietra, A. M. Phanquinone: a useful fluorescent pre-chromatographic derivatization reagent for liquid chromatographic analyses of amino acid dosage forms. Anal. Chim.Acta 2002, 474, 11-20.
10. Gatti, M.; Gioia, M. G.; Andreatta, P.; Pentassuglia, G. HPLC-fluorescence determination of amino acids in pharmaceuticals after pre-column derivatization with phanquinone. J. Pharm. Biomed. Anal. 2004, 35,339-348.
11. Uchiyama, S.; Takehira, K.; Kohtani, S.; Imai, K.; Nakagaki, R.; Tobita, S.; Santa, T. Fluorescence on off switching mechanism of benzofurazans. Org. Biomol. Chem. 2003, 1, 1067-1072.
12. Fillipis, V.; Boni, S.; Dea, E.; Dalzoppo, D.; Grandi, C.; Fontana, A. Incorporation of the fluorescent amino acid 7-azatrytophan into core domain 1-47 of hirudin as a probe of hirudin folding and thrombin recognition. Protein Sci. 2004, 13, 1489-1502.
13. Chersi, A.; Modugno, F.; Rosano, L. Selective “in synthesis” labeling of peptides by fluorochromes.Biochim. Biophys. Acta 1997, 1336, 83-88.
14. Nitz, M.; Mezo, A. R..; Ali, M. H.; Imperali, B. Enantioselective synthesis and application of the highly fluorescent and environment-selective amino acid 6-(2-dimethyl aminonaphthoyl) alanine (DANA).Chem. Commun. 2002, 1912-1913.
15. Cohen, B. E.; McAnaney, T. B.; Park, E. S.; Jan, Y. N.; Boxer, S. G.; Jan, L. Y. Probing protein electrostatics with a synthetic fluorescent amino acid. Science 2002, 296, 1700-1702.
16. Vazquez, M. E.; Nitz, M. E.; Vogel, E. M.; Imperiali, B. Fluorescent caged phosphoserine peptides as probes to investigate phosphorylation-dependent protein associations. J. Am. Chem. Soc. 2003, 125,10150-10151.