Click Here To Get Powerpoint Presentation
Abstract:
Hypertension is a major risk factor for cardiovascular diseases. It is estimated that one billion people in the entire world are affected by high blood pressure [1]. Moreover, the condition is complicated by multiple pathways present in the body which act to maintain blood pressure. Current agents directed toward reduction in the blood pressure are less than satisfactory for patients to reach therapeutic goals (<30% of the patients actually achieve treatment goals). Thus, there is still an ever growing need for new classes of antihypertensive drugs with novel mechanisms that can help in mediating the blood pressure [2].
In the year 1898, Tigerstedt and Bergman showed that kidney extracts from rabbits when injected in other rabbits induced a hypertensive state, thus showing the presence of a pressor agent in the kidney, which they called ‘renin’ [3]. However, pure renin could not be isolated for another 70 years, till Inagami was successful in isolating hog renin and then human renin [4].
Renin is an enzyme that plays a major role in the Renin-Angiotensin System, a regulatory system in the body, which is responsible to maintain homeostasis of blood pressure. The enzyme belongs to the family of aspartic proteases and is responsible for the conversion of inactive angiotensinogen to angiotensin I (Ang I). Angiotensin I by itself is inactive. However, when acted upon by angiotensin converting enzyme (ACE) it gets converted to angiotensin II, which is active and is responsible for most of the pressor effects(Figure 1). Conversion of angiotensinogen to angiotensin I is the rate determining step of the system. The catalytic role played by renin is thus crucial in mediating blood pressure by the Renin-Angiotensin System [5].
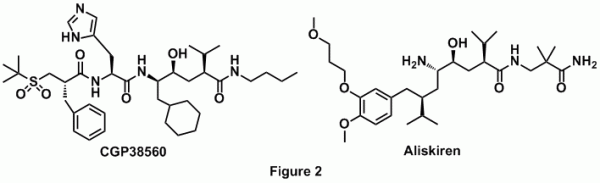
Direct renin inhibition offers another weapon in the arsenal against hypertension. Early inhibitors of renin were monoclonal antibodies, which were excellent probes of enzyme function [6]. However, they were in no ways suitable for use as medication as most were immunogenic and had to be administered via parenteral route. Transition state analogs in the form of statins were first synthesized and were found to be potent inhibitors of renin. However, they had drawbacks because of their peptide like nature and their lack of oral bioavailability [7]. Modifications of these statins led to the development of CGP38560, a compound with reduced peptidic character and of smaller size (MW=730). Optimization of this compound by Novartis led to the development of Aliskiren- the only direct renin inhibitor which is clinically used as an antihypertensive drug (Figure 2) [8, 9, 10].
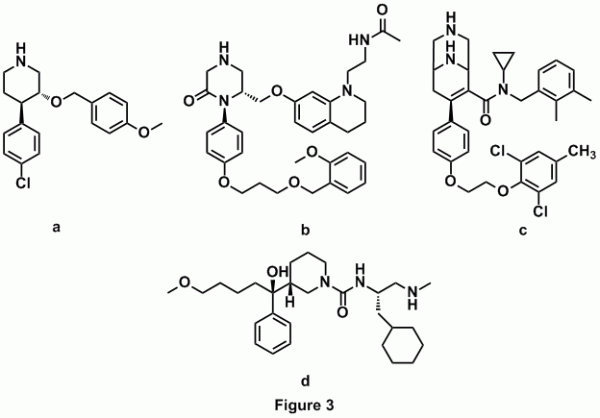
While Novartis was developing inhibitors by modification of the peptide-like inhibitors of renin, Hoffman-La Roche started developing renin inhibitors, which were completely different in structure. Screening of the Roche compound libraries led to the identification of rac-2 which was selective in inhibiting renin over other aspartic proteases (Figure 3.a) [11]. Hoffman-La Roche pursued the development of these compounds until 2001 advancing to pre-clinical stage [12, 13]. Based on the piperidine structure, Pfizer pursued the task of designing ketopiperazine-based renin inhibitors which have shown greater potential (Figure 3.b) [14, 15]. More recently a new series of renin inhibitors based on the ketopiperazine structure was developed by Actelion Pharmaceuticals. These molecules have a 3,9-diazabicyclo[3.3.1] nonene group in place of the ketopiperazine group (Figure 3.c) [16]. Another group of chemists from Vitae Pharmaceuticals has developed orally bioavailable alkyl amines based solely on a computational structure-based design (Figure 3.d) [17].
Today, the only renin inhibitor which is used as an antihypertensive drug in the market is Aliskiren (Novartis). However, it is most probable that new antihypertensive drugs will soon reach the clinic in the near future. The regenerated interest in developing direct renin inhibitors has pushed the design of this class of antihypertensive drugs more than ever before.
Update
In 2011, Aliskiren was found to induce non fatal stroke, renal complications, hypotension and hyperkalemia in patients with diabetes and renal failure.[18] Such contraindications were also found in case of use of aliskiren along with other drugs targeting the RAS. As a result of which a contraindication was added to the label of aliskiren as recommended by the european medicines agency concerning the use along with angiotensin receptor blockers and ACE inhibitors in patients with diabetes or with renal failure.[19]
References:
- Yokokawa, F.; Maibaum, J. Recent advances in the discovery of non-peptidic direct renin inhibitors as antihypertensives: new patent applications in years 2000-2008. Expert Opin. Ther. Patents 2008, 18, 581-602.
- Kearney, P. M.; Whelton, M; Reynolds, K.; Muntner, P.; Whelton, P.; He, J. Global burden of hypertension: analysis of worldwide data. Lancet 2005, 365, 217-223.
- Phillips, M. I.; Schmidt-Ott, K. M. The discovery of renin 100 years ago. News Physiol. Sci. 1999, 14, 271-274.
- Inagami, T. A memorial to Robert Tiegerstedt: The centennial of renin discovery. Hypertension 1998, 32, 953-957.
- The Renin-Angiotensin System; Robertson, J. I. S., Nicholls, M. G., Eds.; Mosby: London, 1993; 2 vols.
- Galen, F. X.; Devaux, C.; Atlas, S.; Guyenne, T.; Menard, J.; Corvol, P.; Simon, D.; Cazaubon, C.; Richer, P.; Badouaille, G.; Richaud, J. P.; Gros, P.; Pau, B. New monoclonal antibodies directed against human renin: Powerful tools for the investigation of the renin system. J. Clin. Invest. 1984, 74, 723-735.
- Lunney, E. A.; Humblet, C. Renin inhibitor design through molecular modeling. In Medicinal Chemistry of The Renin-Angiotensin System. Timmermans, P. B.; Wexler, R. R. Eds.; Elsevier : New York, 1994; 21, pp 73-102.
- Goshke, R.; Cohen, N. C.; Wood, J. M.; Maibaum, J. Design and synthesis of novel 2,7-dialkyl substituted 5(S)-amino-4(S)-hydroxy-8-phenyl-octanecarboxamides as in vitro potent peptidomimetic inhibitors of human renin. Bioorg. Med. Chem. Lett. 1997, 7, 2735-2740.
- Goshke, R.; Stutz, S.; Rasetti, V.; Cohen, N. C.; Rahuel, J.; Rigollier, P.; Baum, H. P.; Forgiarini, P.; Schnell, C. R.; Wagner, T.; Gruetter, M. G.; Fuhrer, W.; Schilling, W.; Cumin, F.; Wood, J. M.; Maibaum, J. Novel 2,7-dialkyl substituted 5(S)-amino-4(S)-hydroxy-8-phenyl-octanecarboxamides transition state peptidomimetics are potent and orally active inhibitors of human renin. J. Med. Chem. 2007, 50, 4818-4831.
- Maibaum, J.; Stutz, S.; Goshke, R.; Rigollier, P.; Yamaguchi; Cumin, F.; Rahuel, J.; Baum, H. P.; Cohen, N. C.; Schnell, C. R.; Fuhrer, W.; Gruetter, M. G.; Schilling, W.; Wood, J. M. Structural modifications of the P2’ Position of 2,7-dialkyl substituted 5(S)-amino-4(S)-hydroxy-8-phenyl-octanecarboxamides: The discovery of aliskiren, a potent nonpeptide human renin inhibitor active after once daily dosing in marmosets. J. Med. Chem. 2007, 50, 4832-4844.
- Vieira, E.; Binggeli, A.; Breu, V.; Bur, D.; Fischli, W.; Guller, R.; Hirth, G.; Marki, H. P.; Muller, M.; Oefner, C.; Scalone, M.; Stadler, H.; Wilhelm, M.; Wostl, W. Substituted piperidines- highly potent renin inhibitors due to induced fit adaptation of the active site. Bioorg. Med. Chem. Lett. 1999, 9, 1397-1402.
- Guller, R.; Binggeli, A.; Breu, V.; Bur, D.; Fischli, W.; Hirth, G.; Jenny, C.; Kansy, M.; Montavon, F.; Muller, M.; Oefner, C.; Stadler, H.; Vieira, E.; Wilhelm, M.; Wostl, W.; Marki, H. P. Piperidine-Renin Inhibitors: compounds with improved physicochemical properties. Bioorg. Med. Chem. Lett. 1999, 9, 1403-1408.
- Marki, H. P.; Binggeli, A.; Bittner, B.; Bohner-Lang, V.; Breu, V.; Bur, D.; Coassolo, P.; Clozel, J. P.; D’Arcy, A.; Doebeli, H.; Fischli, W.; Funk, C.; Foricher, J.; Giller, T.; Gruninger, F.; Guenzi, A.; Guller, R.; Hartung, T.; Hirth, G.; Jenny, C.; Kansy, M.; Klinkhammer, U.; Lave, T.; Lohri, B.; Luft, F. C.; Mervaala, E. M.; Muller D. N.; Muller, M.; Montavon, F.; Oefner, C.; Qiu, C.; Reichel, A.; Sandwald-Ducray, P.; Scalone, M.; Schleimer, M.; Schmid, R.; Stadler, H.; Treiber, A.; Valdenaire, O.; Vieira, E.; Waldmeier, P.; Wiegand-Chou, R.; Wilhelm, M.; Wostl, W.; Zell, M.; Zell, R. Piperidine renin inhibitors: from leads to drug candidates. Il Farmaco 2001, 56, 21-27.
- Holsworth, D. D.; Powell, N. A.; Downing, D. M.; Cai, C.; Cody, W. L.; Ryan, M.; Ostroski, R.; Jalaie, M.; Bryant, J. W.; Edmunds, J. J. Discovery of novel non-peptidic ketopiperazine-based renin inhibitors. Bioorg. Med. Chem. 2005, 13, 2657-2664.
- Holsworth, D. D.; Cai, C.; Cheng, X. M.; Cody, W. L.; Downing, D. M.; Erasga, N.; Lee, C.; Powell, N. A.; Edmunds, J. J.; Stier, M.; Jalaie, M.; Zhang, E.; McConnell, P.; Ryan, M. J.; Bryant, J.; Li, T.; Kasani, A.; Hall, E.; Subedi, R.; Rahim, M.; Maiti, S. Ketopiperazine-based renin inhibitors: Optimization of the “C” ring. Bioorg. Med. Chem. Lett. 2006, 16, 2500-2504.
- Bezencon, O.; Bur, D.; Weller, T.; Bildstein, S. R.; Remen, L.; Stifferlen, T.; Corminboeuf, O.; Grisostomi, C.; Boss, C.; Prade, L.; Delahaye, S.; Treiber, A.; Strickner, P.; Binkert, C.; Hess, P.; Steiner, B.; Fischli, W. Design and preparation of potent, nonpeptidic, bioavailable renin inhibitors. J. Med. Chem. 2009, 52, 3689-3702.
- Tice, C. M.; Xu, Z.; Yuan, J.; Simpson, R. D.; Cacatian, S. T.; Flaherty, P. T.; Zhao, W.; Guo, J.; Ishchenko, A.; Singh, S. B.; Wu, Z.; Scott, B. B.; Bukhtiyarov, Y.; Berbaum J.; Mason, J.; Panemangalore, R.; Cappiello, M. G.; Muller, D.; Harrison, R. K.; McGeehan, G. M.; Dillard, L. W.; Baldwin, J. J.; Claremon, D. A. Design and optimization of renin inhibitors: Orally bioavailable alkyl amines. Bioorg. Med. Chem. Lett. 2009, 19, 3541-3545.
- Parving, H.H.; Brenner, B.M.; McMurray, J.J.; de Zeeuw, D.; Haffner, S.M.; Solomon, S.D.; Chaturvedi, N.; Persson, F.; Desai, A.S.; Nicolaides, M.; Richard, A.; Xiang, Z.; Brunel, P.; Pfeffer, M.A.; ALTITUDE Investigators. Cardiorenal end points in a trial of aliskiren for type 2 diabetes. New Engl. J. Med. 2012, 367, 2204-2213.
- European Medicines Agency – Press Release: 17 February 2012, EMA/CHMP/112042/2012. Address: http://www.ema.europa.eu/docs/en_GB/document_library/Press_release/2012/02/WC500122913.pdf. (Accessed on Feb, 15, 2014).


Awesome! Thanks your sharing!
Hey There. I found your blog using msn. This is
a very well written article. I will make sure
to bookmark it and return to read more of your useful information. Thanks for the post.
I’ll certainly comeback.
hey..its really very good!!… but I need the figures you used because I have aresearch to do about Aliskiren and the figures in your presentation are very useful I understand the SAR from it so can you send me the figures or show me the way to find such ones OK thaanx in advance 😀
Hi samarz,
Glad my presentation was useful to you. You can use the figures directly from the presentation by using print screen option. However, whenever you do use any image taken from it please do mention in your report/ presentation that you have taken it from PharmaXChange.info. Proper credit should be given to the website to prevent plagiarism.
Regards.
very good