Electrophoresis is a analytical technique which separates molecules on the basis of its mass to charge ratio. Gel electrophoresis utilizes a gel as a sieving and anti-convective medium in order to separate molecules and proteins. In gel electrophoresis, an electric field is applied across the gel. Charged molecules such as DNA and proteins begin to migrate across the gel in the presence of an electric field. The rule of movement in gel electrophoresis is larger molecules move more slowly, while smaller molecules move through the gel quickly. Also higher charged species move down the gel more quickly as they will be attracted by the opposite electric field. This difference in speeds between molecules causes them to separate out on the gel (similar to cars on a race track who will separate as the race goes on based on their performance). Finally different bands of different sized and charged molecules will form on the gel.
Gel electrophoresis is widely used in forensics, molecular biology, genetics, and biotechnology. It has been widely used to analyze nucleic acids and proteins. The use of SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis) type of gel electrophoresis is utilized in the analysis of proteins. In this technique sodium dodecyl sulfate acts as a dissociating agent in order to separate proteins into polypeptide chains. This detergent wraps around the polypeptide chain in a constant weight proportion, thus making the intrinsic charges of the peptides negligible, and thus the movement of the peptides will be independant of charge, and be only a linear function of their logarithm of molecular weights. The animation below shows how gel electrophoresis can be utilized to separate nucleic acids.
The animation was provided by the DNA Learning Center and their YouTube Channel. “The mission of the DNA Learning Center is to prepare students and families to thrive in the gene age.”
Animation
[swfobject]1486[/swfobject]
Transcript
In the early days of DNA manipulation, DNA fragments were laboriously separated by gravity. In the 1970s, the powerful tool of DNA gel electrophoresis was developed. This process uses electricity to separate DNA fragments by size as they migrate through a gel matrix.
Gel electrophoresis can be used to separate DNA fragments. Electrophoresis uses an electric current to separate different-sized molecules in a porous, sponge-like matrix. Smaller molecules move more easily through the gel pores than larger molecules.
While at Cold Spring Harbor Laboratory, Phil Sharp, Joe Sambrook and Bill Sugden developed the DNA electrophoresis technique using an agarose gel, made from highly purified seaweed. This could be used to separate DNA molecules ranging from several hundred nucleotides in length to over 10,000 nucleotides.
The gel is submersed in a tank filled with a salt solution that conducts electricity.
Using a pipette, DNA samples are loaded into slots made in the agarose gel. The DNA samples are colorless, but researchers add a blue “tracking” dye. This makes it easier to load the samples, and visually track the DNA migration through the gel.
The phosphate groups in the DNA backbone carry negatively-charged oxygens-giving a DNA molecule an overall negative charge. In an electric current, the negatively-charged DNA moves toward the positive pole of the electrophoresis chamber.
The DNA molecules move though the gel by “reptation”- a reptile-like snaking action through the pores of the agarose matrix. Smaller DNA fragments migrate faster and further over a given period of time than do larger fragments. This is how DNA fragments can be separated by size in an agarose gel.
Sharp, Sambrook and Sugden introduced the use of the fluorescent dye, ethidium bromide, to stain DNA. Ethidum bromide binds tightly to the DNA double helix, and glows when illuminated with ultraviolet light. This lets researchers see where the separated DNA fragmented end up. A photo of the gel can be taken for later analysis.

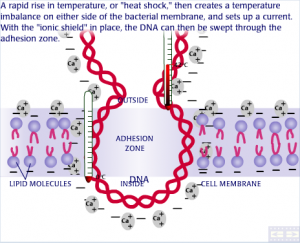

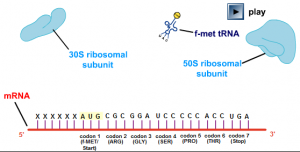
Oh,,This very good and Interesting to understand about gel electrophoresis to me.