Tetrahydrofolate is a co-factor which is needed for the de novo synthesis of thymidine monophosphate, which is required for the biosynthesis of bacterial DNA and RNA. Sulfonamides (such as sulfamethoxazole) and diaminopyrimidines (such as trimethoprim) inhibit different enzymes in the biosynthesis of tetrahydrofolate in the bacteria. Due to the inhibited production of tetrahydrofolate, the bacteria is unable to synthesize the thymidine, and is therefore also unable to produce new DNA or RNA. This eventually leads to the death of the bacteria. Both sulfonamides and diaminopyrimidines act by mimicking the substrate of their respective enzymes, and inhibit the enzyme by blocking the active site of the enzyme.
The animation below displays how these drugs compete with the natural substrate of the enzymes and inhibit these enzymes.
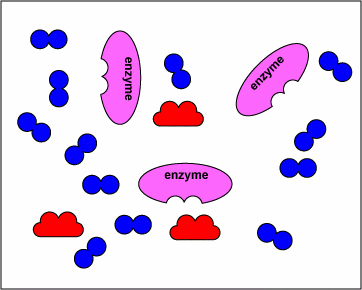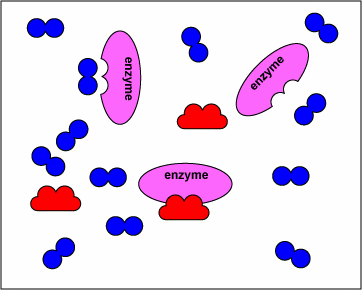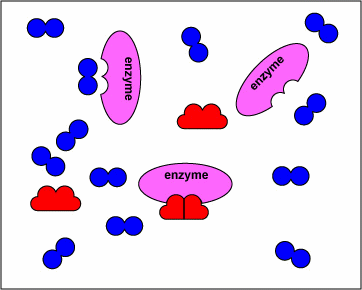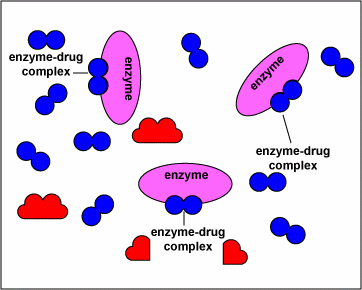
NOTE- The enzymes shown are the targets of sulfonamides and diaminopyrimidines in the biosynthetic pathway of tetrahydrofolate. The substrates are the normal molecules on which these enzymes act. However, in presence of the sulfonamides or the diaminopyrimidines (the drugs) these enzymes get inhibited competitively.
Mechanism of Action
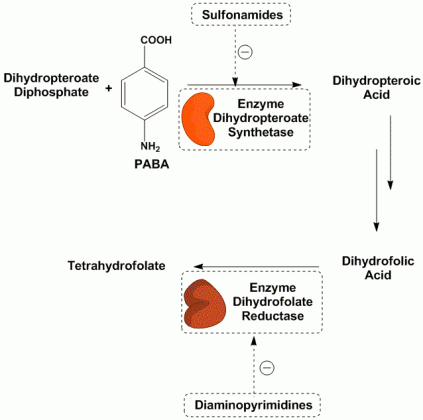
Taking a look at the biochemistry of tetrahydrofolate biosynthesis we can see that tetrahydrofolate is synthesized from the starting material of dihydropteroate diphosphate and p-amino benzoic acid (PABA). These are starting materials which are produced by the bacteria. From these two starting materials, the bacteria attempts to produce dihydropteroic acid. This step is performed by dihydropteroate synthetase. Structurally, sulfonamides mimic the p-amino benzoic acid, and therefore they are capable of going and binding to the active site of the enzyme, thereby competing with the PABA and preventing the synthesis of the dihydropteroate. This eventually leads to a deficiency in the tetrahydrofolate and thymidine causing the bacteria to die.
If however, sulfonamides are not used, the biosynthesis of the tetrahydrofolate continues on till it reaches another check-point, the conversion of dihydrofolic acid to tetrahydrofolate (the final step in the tetrahydrofolate biosynthesis). Here the bacterial enzyme dihydrofolate reductase catalyzes this step. diaminopyrimidines however, mimic the dihydrofolic acid and thus block the active site of this enzyme and block the biosynthesis of the essential tetrahydrofolate leading to bacterial cell death.
Thus, both sulfonamides as well as diaminopyrimidines inhibit bacterial growth by blocking the biosynthesis of tetrahydrofolate, an essential factor needed for the production of thymidine which is required for synthesis of DNA and RNA. While sulfonamides act higher up in the biosynthetic procedure, diaminopyrimidines (such as trimethoprim) act by inhibition of the final step of the biosynthesis. Indeed, since both targets are different but the pathway is the same, these drugs are used as a combination treatment in order to show synergy. The most common combination drug is the cotrimazole combination which is a mixture of sulfamethoxazole (as the sulfonamide) and trimethoprim (as the diaminopyrimidine).
The above animations has been supplied by Dr. Gary E. Kaiser from the Community College of Baltimore County– and it illustrates the mechanism of action of how sulfonamides and diaminopyrimidines act by competitive inhibition of various enzymes in the biosynthesis of tetrahydrofolate. The transcript from his website states:
“The sulfonamides ( sulfamethoxazole, sulfanilamide) and diaminopyrimidines (trimethoprim) block enzymes in the bacteria pathway required for the synthesis of tetrahydrofolic acid, a cofactor needed for bacteria to make the nucleotide bases thymine, guanine, uracil, and adenine. This is done through a process called competitive antagonism whereby a drug chemically resembles a substrate in a metabolic pathway. Because of their similarity, either the drug or the substrate can bind to the substrate’s enzyme. While the enzyme is bound to the drug, it is unable to bind to its natural substrate and that blocks that step in the metabolic pathway. Typically, a sulfonamide and a diaminopyrimidine are combined. Co-trimoxazole, for example, is a combination of sulfamethoxazole and trimethoprim.Sulfonamides such as sulfamethoxazole tie up the first enzyme in the pathway, the conversion of para-aminobenzoic acid to dihydropteroic acid. Trimethoprim binds to the third enzyme in the pathway, an enzyme that is responsible for converting dihydrofolic acid to tetrahydrofolic acid. Without the tetrahydrofolic acid, the bacteria cannot synthesize DNA or RNA.”
References
- Hitchings G. H. Mechanism of Action of Trimethoprim-Sulfamethoxazole—I. Journal of Infectious Diseases. Volume 128 Suppl. 3. S433-S436.

This information was given in a detailed way.its nice and helpful.